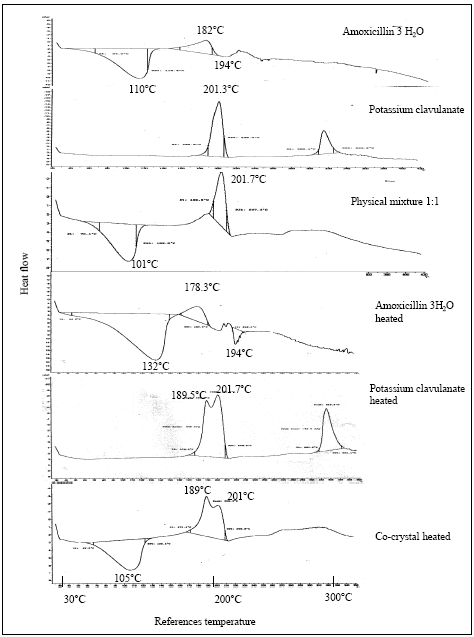
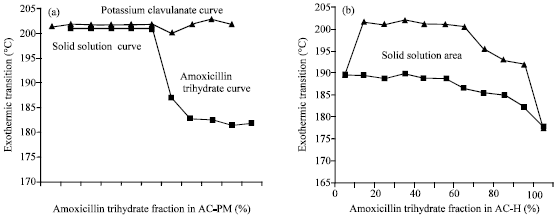
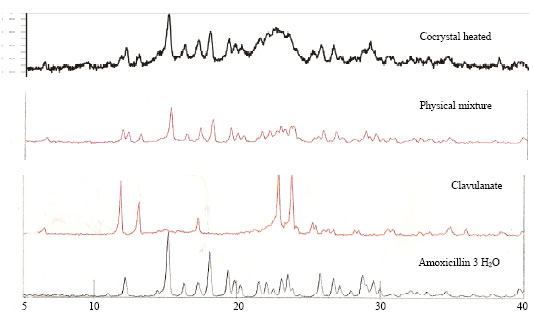
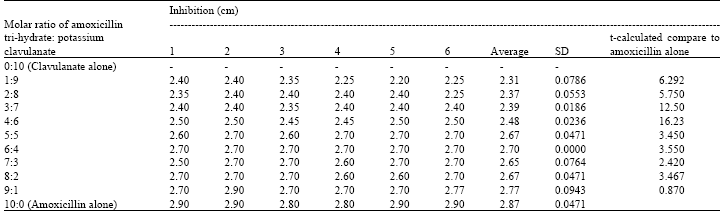
Research Article
The Antibiotic Potency of Amoxicillin-Clavulanate Co-Crystal
Crystallography and Technology Pharmacy Laboratory, Institute Technology Bandung, Indonesia
Sukmadjaja Asyarie
Chemical Pharmacy and Microbiology Laboratory, Institute Technology Bandung, Indonesia
Sundani Nurono Soewandhi
Pharmacokinetics Laboratory, School of Pharmacy, Institute Technology Bandung, Indonesia
Slamet Ibrahim
Pharmacokinetics Laboratory, School of Pharmacy, Institute Technology Bandung, Indonesia