Research Article
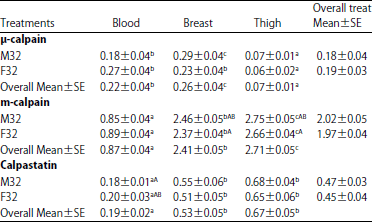
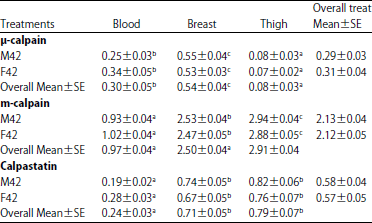
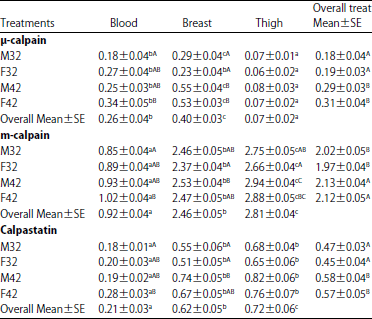
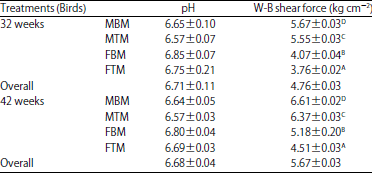
Comparison of Levels of Calpains and Calpastatin in Blood and their Distribution in Skeletal Muscle of Turkey
Division of Post-Harvest Technology, ICAR-Central Avian Research Institute, Izatnagar, 243122 Bareilly, Uttar Pardesh, India
Ashim K. Biswas
Division of Post-Harvest Technology, ICAR-Central Avian Research Institute, Izatnagar, 243122 Bareilly, Uttar Pardesh, India
LiveDNA: 91.394
Samarth Tandon
Division of Post-Harvest Technology, ICAR-Central Avian Research Institute, Izatnagar, 243122 Bareilly, Uttar Pardesh, India
C.K. Beura
Division of Post-Harvest Technology, ICAR-Central Avian Research Institute, Izatnagar, 243122 Bareilly, Uttar Pardesh, India
A.S. Yadav
Division of Post-Harvest Technology, ICAR-Central Avian Research Institute, Izatnagar, 243122 Bareilly, Uttar Pardesh, India