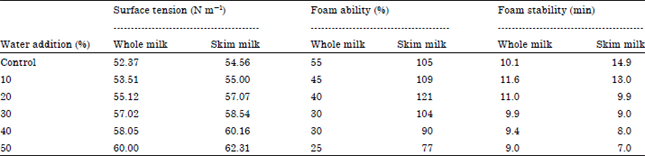
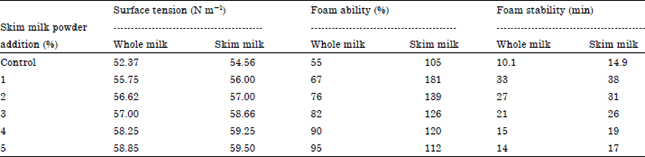
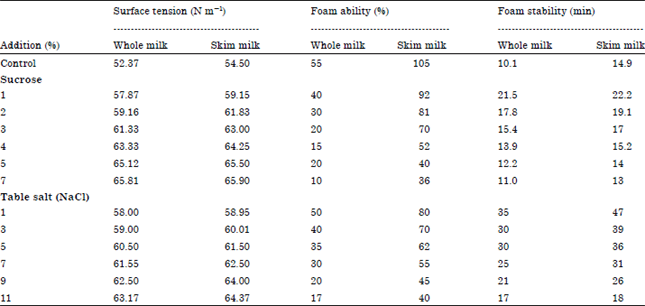
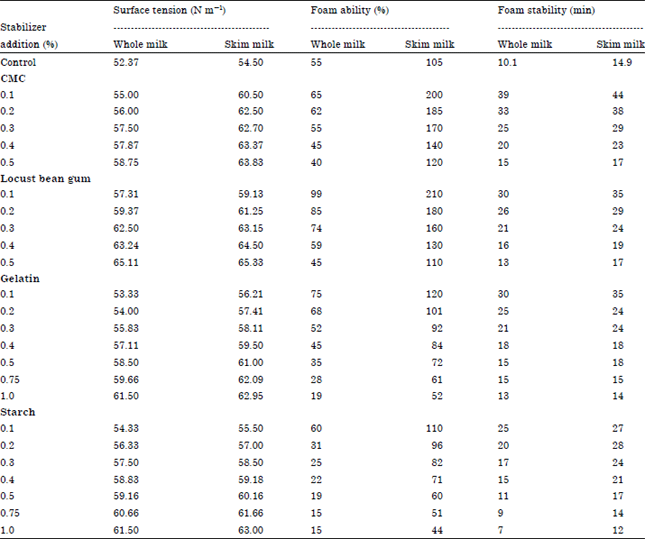
Research Article
Surface Tension and Foaming Properties as a Simple Index in Relation to Buffalo Milk Adulteration
Department of Food Science, Faculty of Agriculture, Ain Shams University, Shoubra Khima, Cairo, 11241, Egypt
Z.M.R. Hassan
Department of Food Science, Faculty of Agriculture, Ain Shams University, Shoubra Khima, Cairo, 11241, Egypt
Wafaa M. Salama
Department of Dairy Technology Research, Food Technology Research Institute, Agricultural Research Center, Giza, Egypt