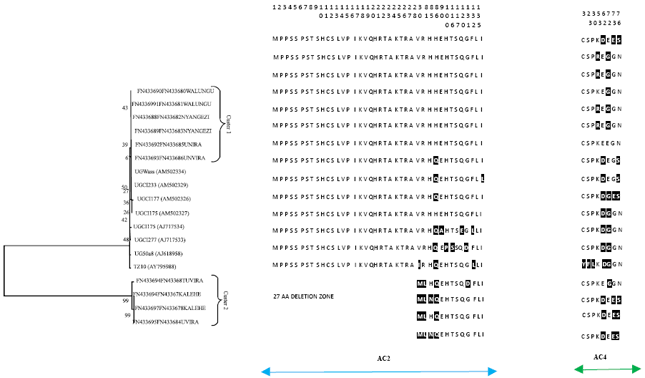
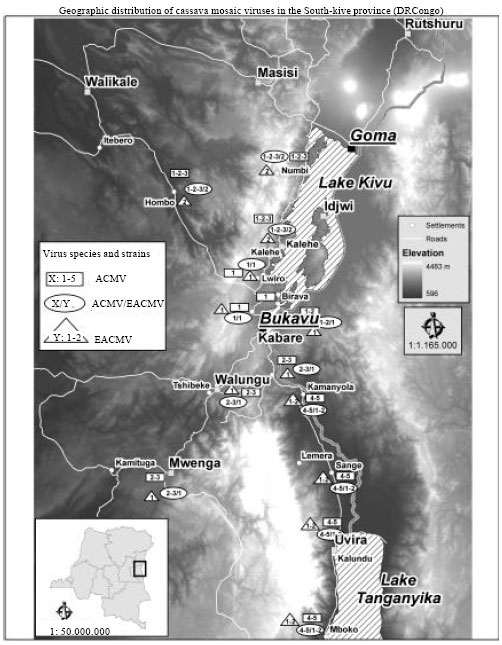
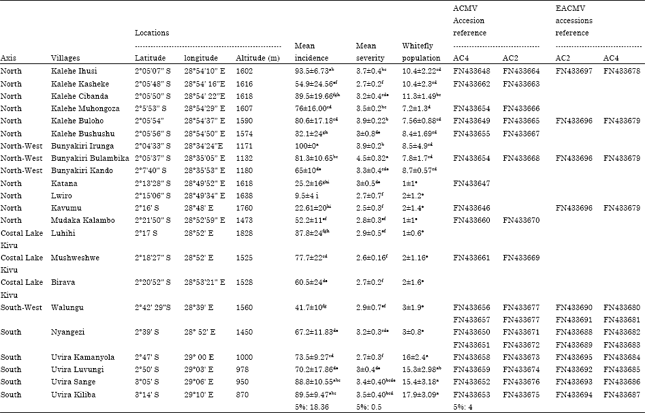
Research Article
Occurrence and Distribution of Cassava Mosaic Begomovirus Related to Agro-ecosystems in the Sud-kivu Province, Democratic Republic of Congo
Earth and Life Institute, Applied Microbiology, Phytopathology, Universite catholique de Louvain, Croix du Sud 2 boite L 07.05.03., 1348 Louvain-la-Neuve/Belgium
J. Walangululu
Faculte des Sciences Agronomiques, Universite Catholique de Bukavu, BP 285 Bukavu, Democratic Republic of Congo
C. Bragard
Earth and Life Institute, Applied Microbiology, Phytopathology, Universite catholique de Louvain, Croix du Sud 2 boite L 07.05.03., 1348 Louvain-la-Neuve/Belgium