Research Article
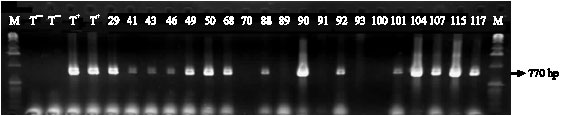
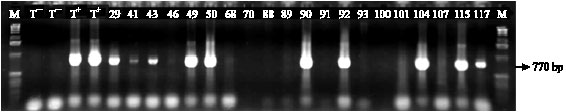
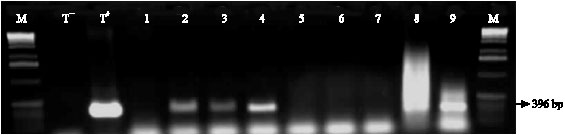
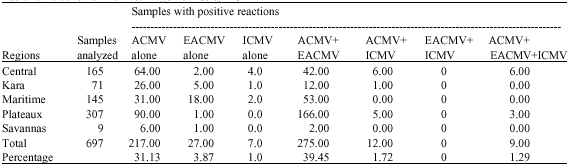
Incidence of Cassava Viral Diseases and First Identification of East African cassava mosaic virus and Indian cassava mosaic virus by PCR in Cassava (Manihot esculenta Crantz) Fields in Togo
Laboratoire de Virologie et de Biotechnologie Vegetales (LVBV), Ecole Superieure d`Agronomie, Universite de Lome, B.P. 1515, Lome, Togo
E. Muller
CIRAD-UMR BGPI (Biologie et Genetique des Interactions Plantes-Parasites) TA41/K, Campus International de Baillarguet, 34398 Montpellier Cedex 5, France
M. Aziadekey
Laboratoire de Virologie et de Biotechnologie Vegetales (LVBV), Ecole Superieure d`Agronomie, Universite de Lome, B.P. 1515, Lome, Togo
Y.M.D. Gumedzoe
Laboratoire de Virologie et de Biotechnologie Vegetales (LVBV), Ecole Superieure d`Agronomie, Universite de Lome, B.P. 1515, Lome, Togo
M. Peterschmitt
CIRAD-UMR BGPI (Biologie et Genetique des Interactions Plantes-Parasites) TA41/K, Campus International de Baillarguet, 34398 Montpellier Cedex 5, France