Short Communication
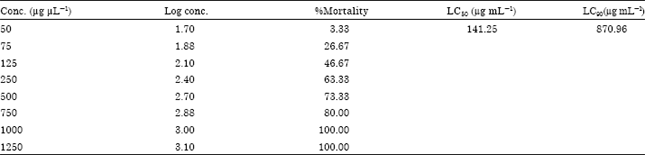
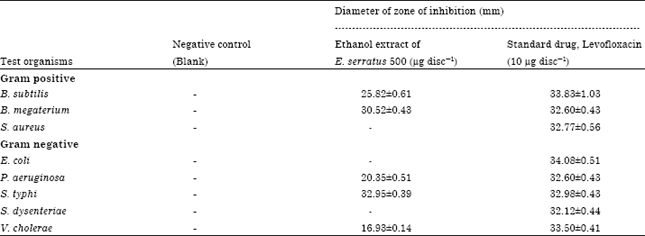
Phytochemical Investigation with Assessment of Cytotoxicity and Antibacterial Activities of the Ethanol Extract of Elaeocarpus serratus
Department of Pharmacy, BGC Trust University, Bangladesh
A. Chowdhury
Department of Pharmacy, BGC Trust University, Bangladesh
J. Das
Department of Pharmacy, BGC Trust University, Bangladesh
A. Chowdhury
Albion Laboratories Ltd., Chittagong, Bangladesh
S.Z. Raihan
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh
M.A. Muhit
Department of Clinical Pharmacy and Pharmacology, Faculty of Pharmacy, University of Dhaka, Bangladesh