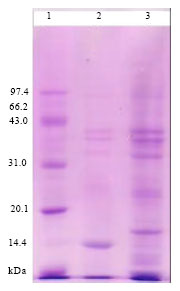
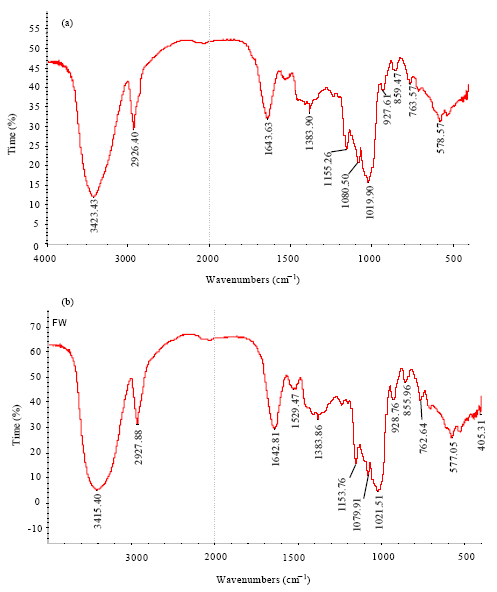
Research Article
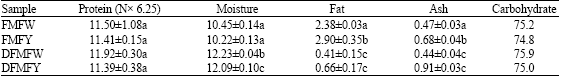
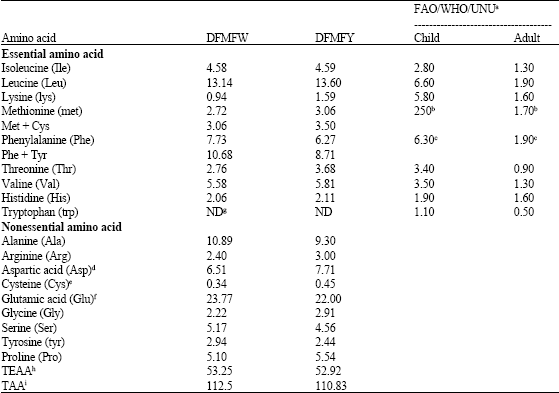
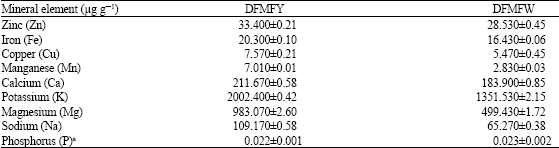
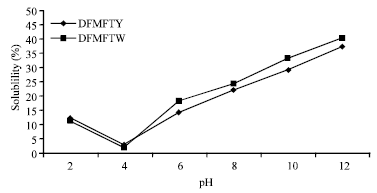
Comparative Study of Chemical Composition and Physicochemical Properties of Two Varieties of Defatted Foxtail Millet Flour Grown in China
State Key Laboratory of Food Science and Technology, Jiangnan University, No 1800, Lihu Road, Wuxi, 214122 Jiangsu, Peoples Republic of China
Z. Huiming
State Key Laboratory of Food Science and Technology, Jiangnan University, No 1800, Lihu Road, Wuxi, 214122 Jiangsu, Peoples Republic of China
Z. Kexue
State Key Laboratory of Food Science and Technology, Jiangnan University, No 1800, Lihu Road, Wuxi, 214122 Jiangsu, Peoples Republic of China
I. Amadou
State Key Laboratory of Food Science and Technology, Jiangnan University, No 1800, Lihu Road, Wuxi, 214122 Jiangsu, Peoples Republic of China
F. Tarawalie
Milton Margai College of Education and Technology, Goderich Campus, Freetown, Affiliated to Njala University, Sierra Leone