Research Article
Effect of Low Temperature, Radiation and their Combination Treatments on Microorganisms Associated with Fresh Water Mola Fish, Amblypharyngodon mola (Hamilton-Buchanan 1822)
Institute of Food and Radiation Biology, Atomic Energy Research Establishment, Savar, Dhaka-1344, Bangladesh
Md. Golam Sorrowar
Department of Zoology, Jahangirnagar University, Dhaka, Bangladesh
Md. Zahid Hasan
Institute of Food and Radiation Biology, Atomic Energy Research Establishment, Savar, Dhaka-1344, Bangladesh
Md. Mansurul Haque
Department of Zoology, Jahangirnagar University, Dhaka, Bangladesh
Harun-Or- Rashid
Institute of Food and Radiation Biology, Atomic Energy Research Establishment, Savar, Dhaka-1344, Bangladesh









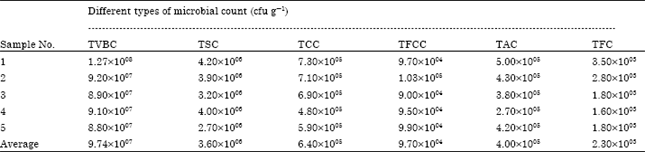
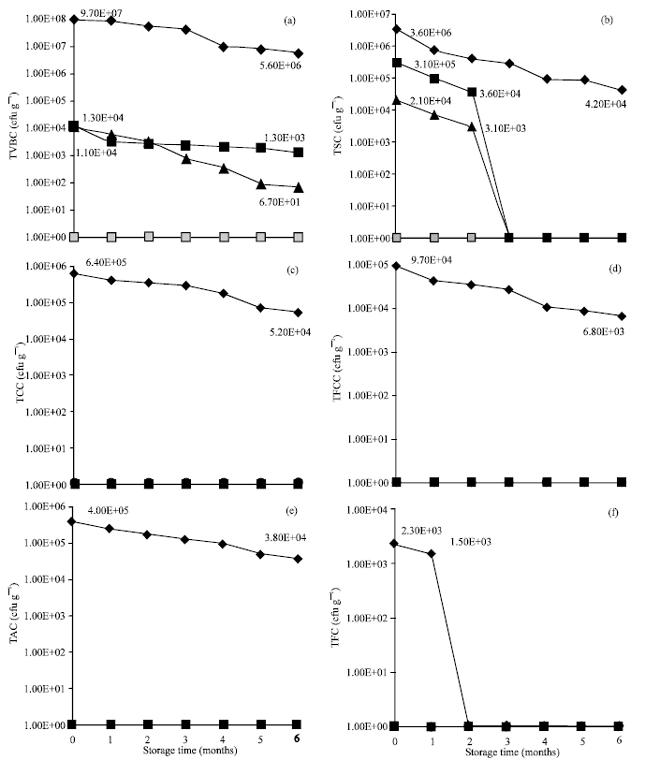

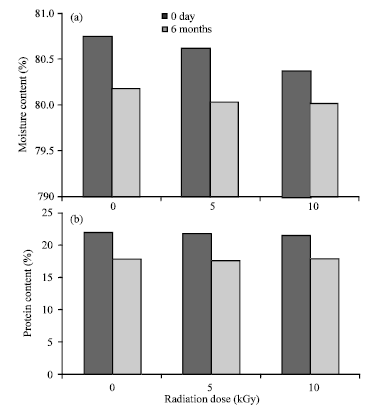
oti jemmimah Reply
pls more short note on ur work with references.