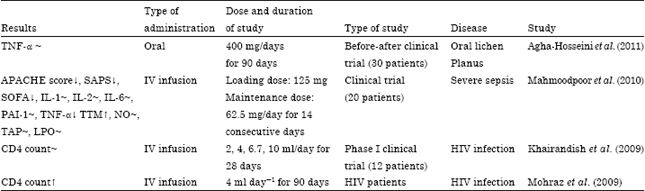
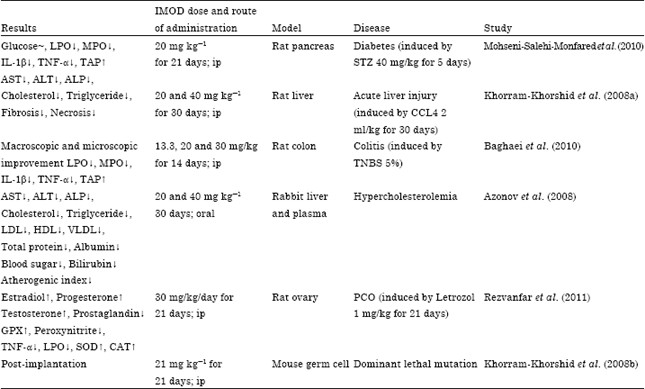
Review Article
Setarud (IMODTM) as a Multiherbal Drug with Promising Benefits in Animal and Human Studies: A Comprehensive Review of Biochemical and Cellular Evidences
Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, Iran
H.R. Khorram-Khorshid
Genetic Research Center, University of Social Welfare and Rehabilitation Sciences, Tehran, Iran
Farhad Gharibdoost
Rheumatology Research Center, Tehran University of Medical Sciences, Tehran, Iran
Mohammad Abdollahi
Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, Iran