ABSTRACT
The specific activity of pyruvate kinase (PK) and phosphoenolpyruvate carboxykinase (PEPCK) of adult I. hypselobagri regulates the glycolysis and CO2 fixation pathway. The PK activity is extremely low, 18.96±0.54 nmol/min/mg proteins, in I. hypselobagri, which is further depleted in in vitro starvation. The PEPCK activity in I. hypselobagri is more or less ten times higher than PK activity, 182±0.1 nmol/min/mg protein, which is further increased in in vitro starvation. The phosphoenolpyruvate (PEP) content of the fresh control fluke varies between 0.532 to 0.608 μmol/mg protein with an average value of 0.556±0.024 μmol/mg protein which is utilized during in vitro starvation. The ratio of PK/PEPCK in fresh fluke is 0.104, which gradually decreases during in vitro starvation period. The PK/PEPCK ratio indicates the CO2 fixation pathway and reverse TCA cycle of energy metabolism is operative in this parasite of swim bladder of Wallago attu.
PDF Abstract XML References
How to cite this article
URL: https://scialert.net/abstract/?doi=jp.2007.51.56
INTRODUCTION
Adult Isoparorchis hypselobagri is a piscine digenetic trematode living in the swim bladder of Wallago attu, an oxygen rich habitat. The metabolic pathway of carbohydrate is not known in this organism. For the present study, enzymes pyruvate kinase (PK) and phosphoenolpyruvate carboxykinase (PEPCK) and substrate phosphoenolpyruvate (PEP) were chosen to study the effects of in vitro starvation in this parasite. PK and PEPCK are assumed to compete for a common substrate, PEP, channeling it to the TCA cycle via pyruvate (PK) and acetyl Co-A or to mitochondrial anaerobic pathways via oxaloacetate (PEPCK) and malate (Saz, 1971; Bryant, 1975). Thus, a ratio of PK and PEPCK is considered as a good indicator of the potential anaerobic capacity of an animal (Simpendorfer et al., 1995).
The present observation deals with the effect of in vitro starvation on key metabolic enzymes PK and PEPCK and key metabolic substrate PEP and assessment of the potential role of increased enzymatic anaerobic potential in adult Isoparorchis hypselobagri.
MATERIALS AND METHODS
Live flukes were collected from the swim bladder of W. attu, obtained from the local fish market and also from the fishermen at Kangsabati reservoir, Mukutmonipur, Bankura, India. Flukes were subjected to starvation for 6,12,18,24,36,48,60 and 72 h in phosphate buffered saline (PBS, pH 7.0; Taylor and Baker, 1978) without glucose. The parasites were maintained alive under aerobic condition at room temperature (30±1°C) in PBS solution with 250 mg Streptomycin and 100000 Unit Penicillin per 100 mL (Srivastava and Gupta, 1977) to prevent bacterial contamination.
For enzyme assay single fluke was homogenized in 2 mL of ice-cold 0.1M Tris-HCl buffer (pH 7.4), using an all glass-Potter-Elvehjem homogenizer at 4°C (Yusufi and Siddiqi, 1978). The sample was centrifuged at 10000 rpm at 0-4°C for 30 min to remove the cell debris. The supernatant thus obtained was used in the enzyme assay.
PK (EC 2.7.1.40) activity was determined in terms of NADH oxidized at 340 nm following Weber et al. (1965). The PEPCK (EC 4.1.1.32) was determined by measuring the oxidation of NADH in the presence of malate dehydrogenase (MDH) at 340 nm following Utter and Kurahashi (1954). Phosphoenolpyruvate (PEP) content was determined following Czok and Lamprecht (1974). The protein content was determined following Lowry et al. (1951). Molar decadic absorption co-efficient (1 x mol-1x mm-1) for NADH and NADPH at temperature of 25 and 30°C are taken for practical use (NADH …… 6.3x102 at 340 nm) (Bergmeyer, 1974).
RESULTS
The specific activity of Pyruvate Kinase (PK) and phosphoenolpyruvate carboxykinase (PEPCK) in fresh control and in vitro starved adult I. hypselobagri are shown in the Fig. 1. The PK activity in control fluke varies from 18.1 to 19.7 nmol/min/mg proteins with an average value of 18.96±0.54 nmol/min/mg protein, whereas, the PEPCK activity varies from 181 to 184 nmol/min/mg proteins with an average value of 182.1±0.1 nmol/min/mg proteins. So, the PEPCK activity is more or less ten times higher than the PK activity. During in vitro starvation period of 6,12,18,24,36,48,60 and 72 h, the PK activity of the fluke decreases slowly and the PEPCK activity increases highly.
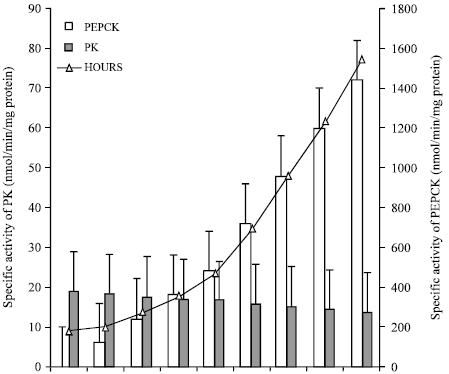 | |
| Fig. 1: | Pyruvate kinase (PK), Phosphoenol pyruvate carboxykinase (PEPCK) activity during in vitro starvation in adult I. hypselobagri |
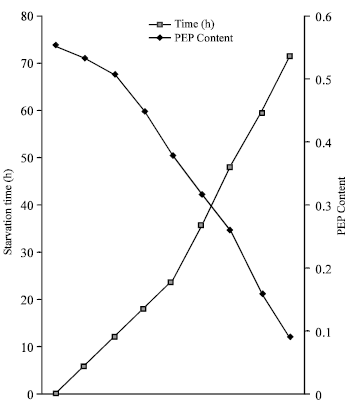 | |
| Fig. 2: | Phosphoenolpyruvate (PEP) content during in vitro starvation in adult I. hypselobagri |
| Table 1: | PK/PEPCK ratio during in vitro starvation in adult I. hypselobagri |
 | |
The PEP is the key metabolic substrate, varies from 0.532 to 0.608 μmol/mg protein with an average value of 0.556±0.024 μmol/mg protein in fresh control fluke which is also decreases during in vitro starvation period (Fig. 2). All the results are highly significant at 0.01% level of significance. The PK/PEPCK ratio in fresh control fluke is 0.104, which decreases during in vitro starvation (Table 1).
DISCUSSION
Two functionally linked enzymes PK and PEPCK involved in this study were used to estimate the potential anaerobic capacity of I. hypselobagri. These two enzymes are likely to compete for a common substrate, phosphoenolpyruvate, channeling it to aerobic (PK) or anaerobic (PEPCK) pathways (Saz, 1971; Bryant, 1975), so that a low PK/PEPCK activity ratio is indicative of a relatively higher anaerobic capacity.
In the present report specific activity of the PK in fresh-control I. hypselobagri is extremely low. Marked differences in the PK activities of helminths are reported mostly on freshly collected cestodes and in few trematodes but there is no report regarding PK activities in starvation condition. In Schistosoma mansoni, male and female shows 2030 (between 1050-2780) and 1150 (between 870-1530) m μmol/min/mg protein, respectively whereas in Schistosoma japonicum male and female shows 1654 (between 1270-2035) and 803 (between 630-1095) m μmol/min/mg protein (Bueding and Saz, 1968), respectively. The low specific activity of PK is also reported from Eurytrema pancreaticum (Vykhrestyuk et al., 1989) and Calicophoron ijimai (Yarygina et al., 1986). Thus, PK appears to catalyze a non-equilibrium reaction and is, therefore, a potential regulatory enzyme. The PK activity, in the present fluke declines during in vitro starvation. There is a possibility of modulators and inhibitors, which regulate the properties of PK activity. Most are activated by fructose-1, 6-bisphosphate in different helminths as in F. hepatica, D. dendriticum, H. contortus, M. expansa, Ligula intestinalis and Litomosoides carinii (Barrett, 1981; Mc Manus, 1975). The ATP and malate also act as inhibitors on PK activity in F. hepatica, D. dendriticum, L. carinii, M. expansa, L. intestinalis, H. contortus, H. diminuta and bicarbonate, lactate, Ca2+ in H. diminuta (Barrett, 1981), resulting the decrease in the specific activity of PK.
The phosphoenolpyruvate carboxykinase (PEPCK) is a rate-limiting enzyme at the branch point of phosphoenolpyruvate (PEP). In some invertebrates, including helminths it appears to function in the direction of PEP carboxylation (Simpson and Awapara, 1966; Bryant, 1975). The enzyme plays a key role in invertebrate energy metabolism because it is a regulatory terminal branch point enzyme, which directs the flow of carbon from PEP into end products of anaerobic metabolism through PEP-succinate pathway. In parasitic helminths, PEPCK catalyzes the reverse reaction, that is, oxaloacetate formation, rather than PEP formation as in mammals (Barrett, 1981). Most of the study on PEPCK activity was based on fresh helminths rather than starved condition. PEPCK has been shown to be active in Fasciola hepatica (Prichard, 1976, 1980) and Schistosoma mansoni (Bueding and Saz, 1968). The report on PEPCK activity is available in Schistosoma mansoni male 408 (367- 483) and in female 109 (102-142) m μmol/min/mg protein (Bueding and Saz, 1968). The PEPCK in helminths catalyzes oxaloacetate formation rather than PEP production (in contrast to mammalian enzyme) and that the enzyme is probably regulatory, with its possible modulators including HCO3-, Mg2+, Mn2+, lactate, GTP, GMP, ATP, ITP, IMP, fumarate, succinate and α-ketogluterate (Smyth and Mc Manus, 1989). In F. hepatica, PEPCK is shown to be most important route for degradation of glucose (Tielens et al., 1987). However, in S. mansoni, experiments with inhibitors of PEPCK gave no indications that this enzyme is involved in the degradation of glucose. In Calicophoron ijimai, high activity of PEPCK but relatively low activities of PK have been reported (Vykhrestyuk and Khamatova, 1983; Vykhrestyuk et al., 1984), which supports the present result. In the present fluke I. hypselobagri, the PEPCK activity increases significantly during in vitro starvation period, which may indicates that CO2-fixation involves in formation of oxaloacetate. Then it is reduced to malate by NADH, formed during glycolysis, a process due to the intervention of a very potent MDH (Bueding and Saz, 1968).
Phosphoenolpyruvate (PEP) is the key glycolytic substrate in parasitic helminths. In many helminths, PEP is a branch point, leading either via PK to pyruvate, or via PEPCK to oxaloacetate and the reverse TCA cycle. In most organisms, which are producing lactic acid, PEP is transformed into pyruvate by means of pyruvate kinase. Read (1951) and Bueding and Saz (1968), reported the PEP in the Hymenolepis diminuta. Thus in the fate of phosphoenol pyruvate, helminths differ from vertebrates (Barrett, 1981). The occurrences of significant PEP levels establish the availability of this substrate of PEPCK in the intact I. hypselobagri.
A small amount of PEP is reported from Fasciola hepatica, F. gigantica (Smyth and Halton, 1983). The PEP is found at significant quantities in fresh-control I. hypselobagri, which declines rapidly during in vitro starvation. It may be due to the fact that PEP is utilized in two pathways. The PEP is converted to pyruvate by the action of PK enzyme and then reduced via LDH to lactate and the other, is the production of oxaloacetate by the enzyme PEPCK that fixes carbondioxide. In the present study PEPCK shows higher activity during in vitro starvation period. Thus, in in vitro starvation period PEP is utilized for fixation of carbon dioxide. Another probable cause may be that the PEP is utilized for neoglucogenesis.
The helminths, which produce carbon dioxide, have a partial reversed TCA cycle and the other rely on glycolysis (Barrett, 1981). Most parasitic helminths seem to have the ability of fixation of carbondioxide. However, the PK/PEPCK ratio in different helminths may give an indication as to which is the major pathway in vivo. Generally, parasites that rely primarily on glycolysis have a ratio in the region 2-10; whilst those helminths rely on CO2-fixation have a region of 0.1-0.05 (Barrett, 1981). In the present study, the PK/PEPCK ratio of fresh control fluke is 0.104 and decrease very much in in vitro starvation period, which is indicative of active CO2-fixation pathway and is indicative of a relatively higher anaerobic capacity during in vitro starvation (Table 1). The PK/PEPCK ratio in S. mansoni male is 5.0 and in female is 9.7 (Bueding and Saz, 1968). Later the PK/PEPCK ratio was reported as 5-10 in S. mansoni and 0.25 to 0.4 in Fasciola hepatica (Barrett, 1981).
So, from the present study it can be concluded that though the parasite live within the O2 rich habitat it may have the strong capability of anaerobic respiration and reverse TCA cycle of carbohydrate energy metabolism.
ACKNOWLEDGMENTS
The authors are grateful to the CSIR, New Delhi for grants in the project No.37 (1023)/99/EMR-II and to the Head, Department of Zoology and Biochemistry, University of Calcutta for kindly providing facilities for this work.
REFERENCES
- Bueding, E. and H.J. Saz, 1968. Pyruvate kinase and phosphoenolpyruvate carboxykinase activities of Ascaris muscle Hymenolepis diminuta and Schistosoma mansoni. Comp. Biochem. Physiol., 24: 511-518.
Direct Link - Prichard, R.K., 1976. Regulation of pyruvate kinase and phosphoenolpyruvate carboxykinase activity in adult Fasciola hepatica Trematoda. Int. J. Parasitol., 6: 227-233.
Direct Link - Simpendorfer, R.W., V.M. Vial, D.A. Lopez, M. Verdala and M.L. Gonzalez, 1995. Relationship between the aerobic and anaerobic metabolic capacities and the vertical distribution of three intertidal sessile invertebrates Jehilus cirratus darwin cirripedia Perumytilus purpuratus lamarck bivalvia and Mytilus chilensis hupe bivalvia. Comp. Biochem. Physiol., 111: 615-623.
- Simpson, J.W. and J. Awapara, 1966. The pathway of glucose degradation in some invertebrates. Comp. Biochem. Physiol., 18: 537-548.
Direct Link - Srivastava, M. and S.P. Gupta, 1977. Studies on in vitro survival of Isoparorchis hypselobagri. Z. Parasitenk, 52: 61-68.
PubMedDirect Link - Vykhrestyuk, N.P. and A.Y. Khamatova, 1983. Cytosol malate dehydrogenase in the trematode Calicophoron ijimai and the effect of some anthelmintic drugs on its activity. Parazitologiya, 17: 397-402.
Direct Link - Weber, G., N.B. Stamn and E.A. Fisher, 1965. Insulin Inducer of Pyruvate Kinase. Science, 149: 65-67.
CrossRef








