Research Article
Sucrose as a Crosslinking Modifier for the Preparation of Calcium Alginate Films via External Gelation
Department of Pharmaceutics and Pharmaceutical Technology, College of Pharmacy, Taif University, Taif, Saudi Arabia
Sodium alginate is a natural substance extracted from brown seaweed and chemically composed of sodium salt of water-soluble polysaccharides comprising (1-4)-linked units of R-D-mannuronate (M) and α-L-guluronate (G). In polymer chains, monomers are arranged alternately in GG and MM blocks, together with MG blocks (King, 1983; Grant et al., 1973). Sodium alginate has been widely used in paper, adhesives, textile, paints, ceramics, food and pharmaceutical industries. One interesting application, in the food and pharmaceutical industry, is its use as a film forming material. Sodium alginate has the ability for film formation upon casting or solvent evaporation. The prepared sodium alginate films are usually examined in terms of their rigidity and functionality such as tensile strength, percentage elongation tear strength, water vapor permeability, oxygen permeability, drug release tests and food preservation activity (Wang et al., 2010; Bajdik et al., 2009; Remunan-Lopez and Bodmeier, 1997; Fontes et al., 2011; Singh, 2008).
The prepared films are usually tailored to suit many pharmaceutical or food applications. For example, it can be used as a carrier for buccal drug delivery (Pongjanyakul and Suksri, 2009; Choi and Kim, 2000) or as film coating material for certain food products (Olivas et al., 2007; Rojas-Grau et al., 2007; Maqsood et al., 2012; Daud et al., 2008). However, sodium alginate is rarely used alone in the preparation of the films rather one or two components are added in order to modify the composition and hence improve the film characteristics such as film cohesion properties and film performance. Alginate modification could be attained through its ability to react with multivalent cations. Since alginate films are hydrophilic matrices, the crosslinking process with tri- and divalent cations has been used to improve their water barrier properties, mechanical resistance, cohesiveness and rigidity (Grant et al., 1973; Nakamura et al., 1995). Many divalent cations were found to induce alginate gelation (Wahab et al., 1997; Olivas and Barbosa-Canovas, 2008). The most interesting cation that has been used frequently to prepare cross linked alginate films is the calcium ions (Olivas and Barbosa-Canovas, 2008; Listiarini et al., 2011; Julian et al., 1988).
The cations establish the cooperative association between M and G blocks, resulting in a tridimensional network where, they may pack and be coordinated by junction zones that involve cooperative association of extended segments of the polymer chains. This arrangement is known as the “eggbox” model (Grant et al., 1973). The process of alginate cross-linking can be achieved usually by the external gelation, or the internal gelation or a combination of both techniques (Schoubben et al., 2010). External gelation can be simply obtained by dropping sodium alginate solution in a calcium chloride solution while, the internal gelation strategy consists of the use of water insoluble calcium salt which is initially dispersed in the sodium alginate solution (Schoubben et al., 2010; Liu et al., 2003; Silva et al., 2006).
In the external gelation, due to the fast cross-linking process between alginate and calcium ions, localized gelling areas are produced, affecting the uniformity and quality of films. Thus, the other technique of internal gelation has been proposed to form homogeneous matrices through the slow release of calcium ions from insoluble calcium salt in an acidified medium. However, internal gelation has the disadvantage of difficulty to be applied on large scale and therefore this limits its industrial use (Schoubben et al., 2010). One important use for calcium alginate films is in cells immobilization (Borgio, 2011; Panesar, 2007; Youssef and Al-Omair, 2008; Hara et al., 2008; Premjet and Tachibana, 2004) where, the production of films with high porosity, strong physical and mechanical properties may positively affect the cells productivity.
The main objective of this study was to improve the external gelation technique by the addition of different percentage of sucrose during the crosslinking process. Sucrose was a good candidate because it is inert, water-soluble and alginate compatible.
Materials used: Sodium alginate was purchased from Sigma-Aldrich (USA), with a viscosity of (2000 mPa sec) at 25°C measured by Vibro-viscometer. Sucrose all other chemicals and reagents were of the highest purity grade commercially available.
Film preparation: Alginate films were made by casting procedure. In the first stage, sodium alginate 1% w/v solution was mechanically stirred at 1000 rpm (Ika homogenizer, Germany) for about 20 min to ensure homogeneity. Sucrose was added in the following percentages 0, 1, 2, 3 and 5% w/v. Afterwards, 15±0.2 g solution was poured in plastic Petri dishes (d = 14 cm). The solution was dried in a convection oven (Memmert, Germany) at a temperature of 75°C for 48 h. Thin transparent films of sodium alginates were prepared. To the sodium alginate dry films a 15 mL calcium chloride solution (2% w/v) was rapidly added to the polymer solution at room temperature. The rapid addition of CaCl2 was necessary to resemble a high speed industrial application which enhances the induction of localized gelation on film surface and thus, affecting film homogeneity. The cross-linked alginate films were soaked with CaCl2 for 2 h before vigorously rinsing the films with distilled water. Then, each film was soaked in 200 mL distilled water and put on shaker at 200 stroke min-1 for 48 h. The films were returned back to the Petri dishes and put in oven at 45°C for 24 h.
All films were conditioned at room temperature and 0% RH inside silica gel desiccators for 3 days before characterization. The moisture content of the films therefore was lower than 2%.
Scanning electron microscope (SEM): The surface and cross-section of calcium alginate films were observed via SEM. The films were coated with gold under vacuum and observed with SEM (AMR Leitz 1000A, Germany).
Fourier transform infrared (FTIR) spectroscopy measurements: The calcium alginate thin films were cut into small square pieces 2x1 cm and mounted on a flat piece on DRS 8000A accessory of the FT-IR Prestige-21 (Shimadzu, Japan) where the diffuse reflectance spectra is converted into transmission spectra for comparison purposes. In case of powder samples of sodium alginate and sucrose, the powder samples were mixed with KBR powder and placed in a sample cup and measured.
DSC of calcium alginate films: Weighed amount of around 5 mg of calcium alginate films were put in an open aluminum DSC pan and heated at a scan speed of 10 EC min-1 over a temperature range of 30-300 EC while purging nitrogen gas with a flow rate of 20 mL min-1 in a differential scanning calorimeter (STA 449 F3 Jupiter, Netzsch, Germany) which was previously calibrated against indium.
Moisture sorption isotherm: Chambers with different relative humidity were prepared ranging from 0 to 90% using saturated salt solutions. The salts used were anhydrous silica gel (0% RH), magnesium chloride (33% RH), sodium chloride (75% RH) and barium chloride (90% RH) according to Weast (1975). Samples were kept inside the chambers at 25°C for 1 week or until reaching equilibrium with the environment according to water activity values present in each humid chamber. Samples were weighed and dried at 110°C for 24 h and let to cool in desiccators containing anhydrous calcium sulfate and weighed again. The moisture content was expressed as percent gain in weight w/w (grams of water per gram of dry solid). The experiment was repeated 5 times and proper statistical treatment was conducted.
Diffusion study: The diffusion study was carried out with the calcium alginate films using Franz diffusion cell. The cell consists of two chambers, the donor and the receiver. The donor chamber was open at the top and was exposed to atmosphere. The temperature was maintained at 25±0.5°C. The donor chamber was supplied with conductimeter (Mettler Toledo, Germany) to measure the changes in conductivity with time for 14 mL aqueous medium of 1% NaCl, where NaCl was used as a diffusion solute. The calcium alginate film was previously soaked for 24 h in deionized water prior each experiment and was put as a membrane separating the two chambers. In the receiver chamber 30 mL distilled water was added. The donor and receiver chambers hold together using clamp. The receiver chamber was stirred with magnetic bar operated by magnetic stirrer, to prevent the formation of concentrated drug solution layer below the calcium alginate film. A calibration curve based on conductivity was established using NaCl in concentration range 0.06-1% NaCl solution, where a linear correlation was obtained with a correlation coefficient of 0.9976. The experiment was repeated 5 times and proper statistical treatment was conducted.
Thermomechanical analyzer (TMA): Film elongation property was determined using TMA. The change in film dimensions upon losing its moisture was measured using TMA instrument. All films were soaked in deionized water for 10 min and excess water was removed using tissue papers. Three rectangular strip specimens (width 5 mm, length 20 mm) were cut for each film for testing. The percentage elongation were determined using a TMA analyzer (TMA 402 F1 Hyperion, Netzsch, Germany), operated at tension mode. Sample holder used was fused silica (tension curve). Then sample measurement was reported at same conditions through applying a constant tension force of 1 N and enabling the auto vacuum pump for another 5 min to produce a negative pressure inside the furnace vessel. The experiment was conducted under isothermal conditions at a temperature of 25°C under vacuum and for a time interval of 5 min.
Statistical treatment: The results were expressed as the mean±SD (standard deviation). For comparison, a one-way Analysis of Variance (ANOVA) was applied (Microsoft Office Excel 2003, Microsoft Corporation). A difference was considered statistically significant when the probability value (p) was less than 0.05.
Calcium alginate films were produced by a two-stage method; the first stage includes formation of a homogeneous composition of sodium alginate/sucrose in a dry solid film. Sucrose was added to act as an inert diluent. In the second stage, calcium chloride was added as a crosslinking agent in order to make an external film gelation in the presence of sucrose. Then, sucrose was removed by washing and the film was left to dry. The formed films showed different physical appearance depending on sucrose concentrations used during film preparation. The film formed without sucrose showed the most folded and brittle structures, as shown in Fig. 1.
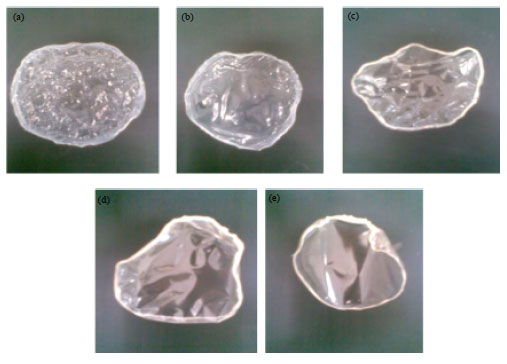 | |
| Fig. 1(a-e): | Calcium alginate films prepared in the presence of different concentration of sucrose 0, 1, 2, 3 and 5% (a, b, c, d and e, respectively), then sucrose was washed out and the films were dried and photographs were taken |
 | |
| Fig. 2(a-e): | Calcium alginate films prepared in the presence of different concentration of sucrose 0, 1, 2, 3 and 5% (a, b, c, d and e, respectively), then sucrose was washed out and the films were dried and SEM photographs were taken for the surface and the cross section of each film |
The addition of limited amount of sucrose in the first stage before the crosslinking process was found to have a positive impact on film homogeneity, transparency, consolidation and visual appearance which could allow the proper handling of the formed films prepared by external gelation. The insertion of intermolecular sucrose prior to crosslinking could partially decrease the degree of irregular polymer crosslinking and it could hamper the rapid crosslinking rates which occurred during the external gelation process.
Surface roughness was visually evaluated using SEM, as shown in Fig. 2 (left-side). The film surface formed without the addition of sucrose appears to be highly irregular and non smooth, while all films formed by the addition of sucrose were more regular and smooth. It was obvious that the surface roughness was almost the same for films formed using sucrose concentrations from 1 up to 3% however at 5% the surface becomes more porous. SEM photos were taken inside the cross section in order to observe the underneath layer physical arrangements, as shown in Fig. 2 (right-side). It is obvious that in the case of film formed without the aid of sucrose, the crosslinking of alginate resulted in the formation of highly crowded layers which could result in polymer folding and abruptly emerging of polymer lumps on the film surface forming inhomogeneous films during the external gelation. However, the addition of sucrose in different concentrations 1-5% could be helpful and it could act as intermolecular inert spacer during the crosslinking of carboxylates with calcium ions and thus the degree and/or the rate of crosslinking might be modified.
In order to investigate the chemical cross linking reaction and the effect of sucrose on the reaction, FTIR scanning was carried out, as shown in Fig. 3. FTIR 2000-400 cm-1 range showed stretching of carboxyl group of sodium alginate at 1607 and 1424 cm-1 which are assigned to symmetric and antisymmetric COO stretching vibrations of the free carboxylate groups as also observed by Bajdik et al. (2009) and Socrates (2001). The addition of calcium ions to carboxyl functional groups of alginate should restrict their movements due to crosslinking and this could explain the shift of wave numbers to higher energy levels 1672 and 1462 cm-1. For films formed by the addition of sucrose, there were no IR peaks related to sucrose which indicated its full removal in the second stage of external gelation. However, the impact of sucrose on film chemical property is still obvious, where, the positions and the shapes of the carboxylate bands indicated a change in the characteristic of the carboxylate groups (Socrates, 2001). The shape and intensities of these bands were largely affected with increasing sucrose content; it can be supposed that the presence of sucrose could help in slowing down the crosslinking interaction as observed in lowering peak wave numbers and band broadness which depends on the amount of sucrose added to the calcium alginate films. In another report, such finding has been also observed after the addition of lactose to alginic acid (Bajdik et al., 2009).
Calcium alginate films were also evaluated by DSC as presented in Fig. 4. The decomposition of the sodium alginate polymer represented by an exothermic peak at 245°C which agrees well with the reported value by Kong et al. (2009). Crosslinking of alginate via calcium in absence of sucrose addition was found to result in a much lower decomposition temperature i.e., 209°C. Besides, the formed calcium alginate films thermograms did not show any peak related to sucrose which indicates its full removal. The addition of sucrose to the calcium alginate films resulted in decomposition temperatures around 220°C. It could be concluded that while sodium alginate decomposes at 245°C, the maximum interaction to produce calcium alginate might be occurred in absence of sucrose and resulted in a decomposition temperature of 209°C. The addition of sucrose could slow down the interactions or could affect the degree of crosslinking which could explain the intermediary decomposition temperature of 220°C for those samples.
The percent moisture sorption of calcium alginate films was dependent on the environmental relative humidity. As relative humidity increases calcium alginate films were able to uptake more water from the environment where the maximum water content was around 40%, as shown in Fig. 5.
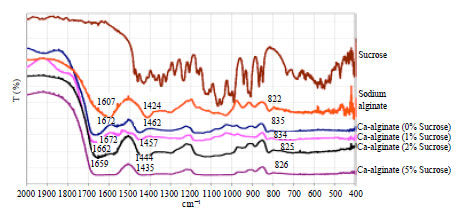 | |
| Fig. 3: | FTIR spectra of sucrose, sodium alginate and calcium alginate films prepared with different sucrose concentrations (0, 1, 2 and 5%) |
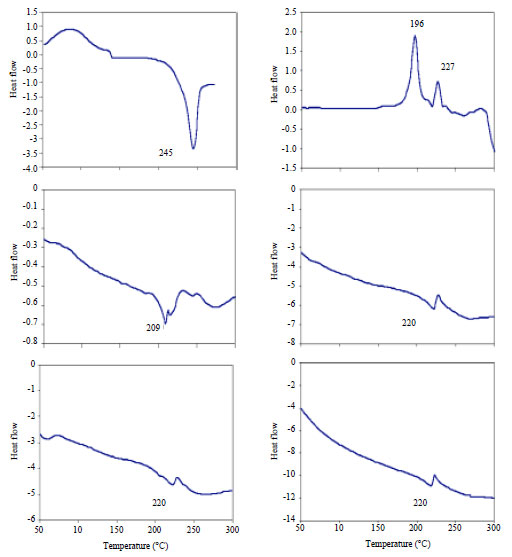 | |
| Fig. 4: | DSC thermograms of sucrose, sodium alginate and calcium alginate films prepared with different sucrose concentrations (0, 1, 2 and 5%) |
However, the trend of increase in moisture content was the same between each films irrespective of the formulation technique i.e., moisture content was not significantly affected (p>0.05) by the amount of sucrose added during formulation rather film moisture content was significantly affected by the relative humidity of the environment (p<0.05). This could indicate that water molecules are easily absorbed and could form an intimate molecular hydration with calcium alginate strands due to high polymer hydrophilicity. Thus, the level of film porosity would not have an important role in polymer hydration property. This agrees well with the previous report of Kamol et al. (2011). The researchers found that calcium alginate could absorb up to 50% of water within a minute and indicated its strong hydrophilic nature.
Film elongation studies are shown in Fig. 6. Determination of the film elongation or shrinkage during drying is an indicator of polymer chain arrangements in the film. Under isothermal conditions and due to the negative pressure produced by the vacuum pump, water is expected to be evaporated out from the calcium alginate films. Since, water provides elasticity of the films its removal on the other hand could result in the increase in film, rigidity, stiffness and degree of shrinkage. In calcium alginate films prepared with zero sucrose, the highly compacted polymer chains having low free intermolecular spaces did not allow elongation rather shrinkage of the film occurred. On the contrary, films prepared with relatively higher percent of sucrose will have higher intermolecular spaces and less tight crosslinking polymers process elongation has been occurred. Consequently, this was dependent on intermolecular spaces which were a result of added sucrose. Calcium alginate films elongation was increasing upon increasing the percent of sucrose added to calcium alginate films.
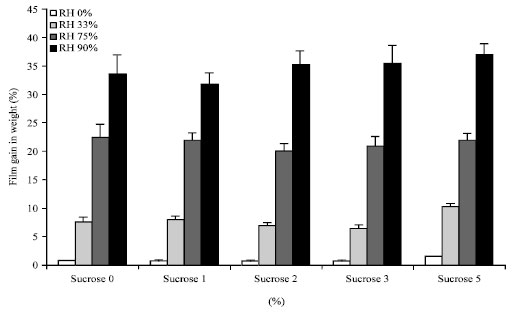 | |
| Fig. 5: | Percentage gain in weight of calcium alginate films exposed to different relative humidity conditions at room temperature and after equilibrium |
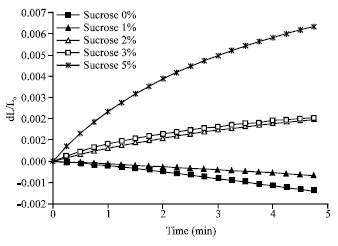 | |
| Fig. 6: | Calcium alginate film elongation- (change in length per initial length dL/Lo) with time after applying a constant force using TMA instrument at room temperature |
Diffusivity of electrolyte solution is expected to be dependent on film porosity which is one important property of the formed calcium alginate films. Calcium alginate film porosity should be dependent on the degree of intermolecular spaces. Figure 7 shows the decrease in conductivity in donor cell upon the transfer of sodium chloride from donor to receiver cell separated by a calcium alginate film. The higher rate of transfer indicates higher permeation ability of sodium chloride which indicates higher porosity or intermolecular spaces. Films formed with higher percent of sucrose were found to have higher rate of sodium chloride transfer.
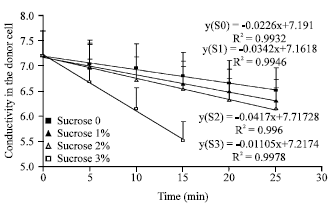 | |
| Fig. 7: | Diffusion of NaCl solution through calcium alginate films measured by Franz cell of diffusion. NaCl solution diffused from donor to receiver chamber separated by calcium alginate films |
On the basis of the results presented here it can be concluded the addition of sucrose during the process of preparation of calcium alginate film, via external gelation, resulted in hampering the crosslinking reaction. This was found to produce a more smooth calcium alginate film which could open the door for the use of such method on large scale production.
The author greatly appreciate the support of Dr. Adnan Badwan (General Director of the Jordanian Pharmaceutical manufacturing Company) on facilitating the conduct of this research.