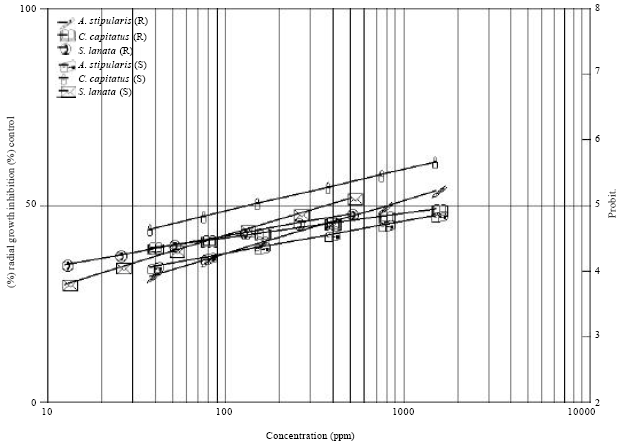
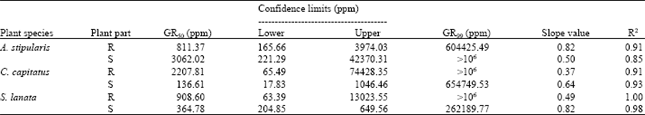
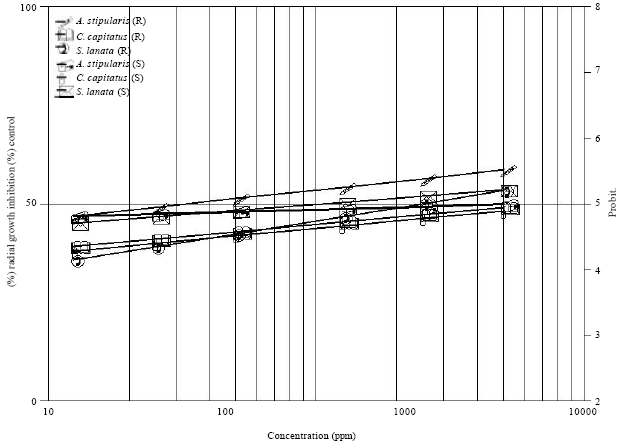
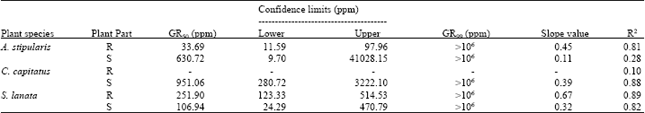
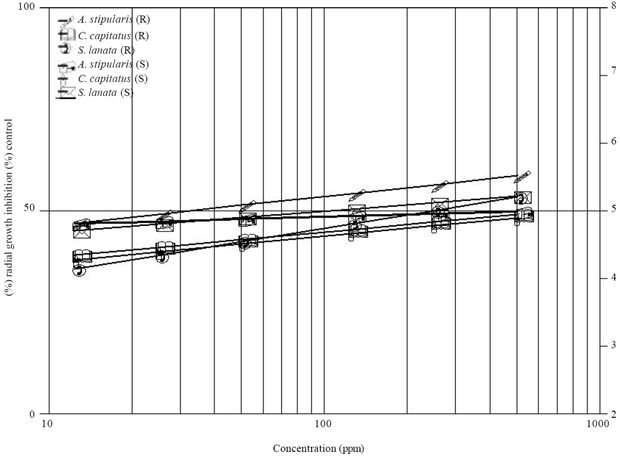
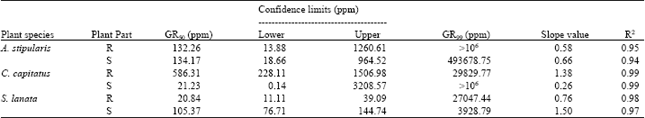
Research Article
In vitro Antifungal Activity of Three Geophytic Plant Extracts against Three Post-harvest Pathogenic Fungi
Department of Agricultural Botany, Faculty of Agriculture, Tanta University, Egypt
Sabry A. Abdallah
Department of Plant Protection, Faculty of Agriculture, Tanta University, Egypt