Research Article
CNS Activity of Methanol and Acetone Extracts of Acorus calamus Leaves in Mice
Department of Pharmacology, College of Medicine and Health Sciences, University of Gondar, P.O. Box 196, Gondar, Ethiopia
N. Jose
Department of Pharmacology, KMCH College of Pharmacy, Coimbatore-641 035, Tamilnadu, India
H. Subhash
Department of Pharmacology, KMCH College of Pharmacy, Coimbatore-641 035, Tamilnadu, India









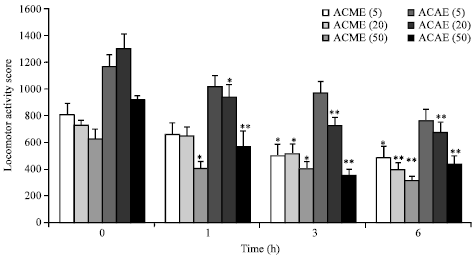
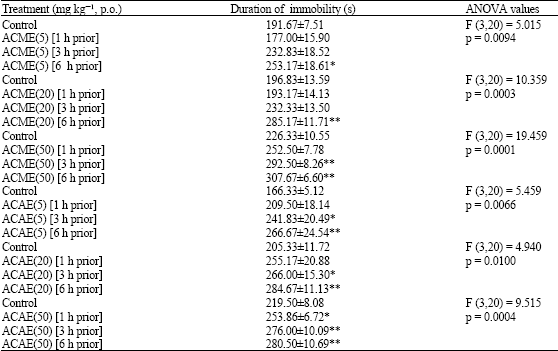

vamsi krishna.a Reply
respected sir's
i would like to know more information about CNS Activity of Acorus calamus Leaves in Mice