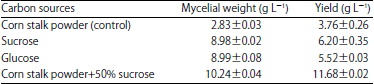Research Article
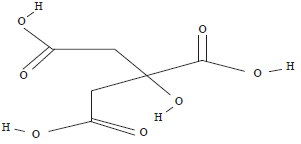
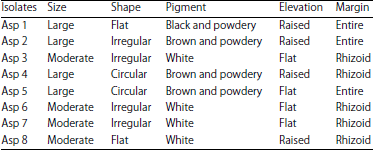
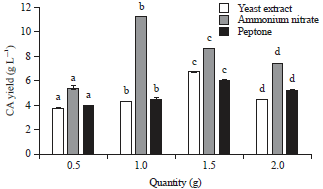
Production of Citric Acid from Corn Stalk through Submerged Fermentation Using Aspergillus niger
Department of Scientific and Industrial Research, National Research Institute for Chemical Technology, Zaria, Nigeria
Shefiat O. Arekemase
Department of Petrochemical and Allied, National Research Institute for Chemical Technology, Zaria, Nigeria
LiveDNA: 234.26890
Ibrahim Abdulwaliyu
Department of Scientific and Industrial Research, National Research Institute for Chemical Technology, Zaria, Nigeria
LiveDNA: 234.25748
Musa Latayo Batari
Department of Scientific and Industrial Research, National Research Institute for Chemical Technology, Zaria, Nigeria
Helen Hoomsuk Raplong
Federal College of Animal Health and Production Technology, Vom, Nigeria
Bamidele D. Aronimo
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria
Yakubu Sani
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria