Research Article
Effect of Threonine Supplementation on Broiler Chicken Productivity Traits
Department of Animal Production, College of Agriculture, University of Baghdad, Baghdad, Iraq
The growth rates of chickens depend on the availability of adequate amount of amino acids that can be used to synthesize proteins involved in many physiological processes, including signal transduction, hormone signaling, cell structure and antioxidant systems1.
Amino acids are also important for energy metabolism, urinary system function and sexual maturation2. Due to the rapid growth of commercial chicken strains, the availability of amino acids for optimal growth, particularly muscle growth as well as physiological function is critical3,4.
In addition to methionine and lysine, the aliphatic amino acid threonine is an important growth factor in broilers5 and is one of the basic amino acids needed for growth of domesticated birds6. Chickens cannot synthesize several amino acids, including threonine and thus threonine supplementation is required7. Amino acids play many important physiological roles8, particularly in thyroid function9 and diet lack of threonine as well as other amino acids can lead to diminished physiological function10-12. Indeed, Azzam et al.13,14 stated that leucine supplementation improved humoral immune responses by increasing γ-globulin levels as well as antioxidant levels in chickens4. Moreover, threonine helps maintain the integrity of the intestinal mucosal barrier and thus can enhance nutrient uptake by broiler chickens15.
Estalkhzir et al.16 found that the addition of threonine to broiler chicken diets increased productivity in terms of Body Weight (BW), Feed Conversion (FC), Dressing Percentage (DP), relative breast weight (BRp) and thigh weight (THp). In addition, Rezaeipour et al.17 reported that threonine supplementation together with feed particle size improved FC, whereas threonine supplementation for the first 42 days after birth improved FC to levels above those seen with the addition of probiotics18.
To date, these studies have not shown a direct role of threonine in improving chicken growth performance, especially in broilers. Therefore, in this study the diets of broiler chickens were supplemented with various threonine concentrations in order to better characterize the role of threonine in growth and productivity traits and to determine the optimal supplementation concentration.
This study was conducted at the poultry farm at the Animal Production Department, College of Agriculture, University of Baghdad, Abu Ghraib, between March 19, 2016 and April 24, 2016.
| Table 1: | Study diet composition |
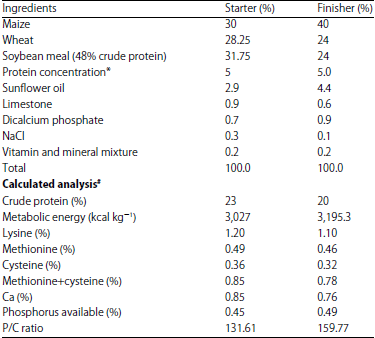 | |
*Brocon-5 Special W: 40% crude protein, 3.5% fat, 1% fiber, 6% Ca, 3% available P, 3.25% lysine, 3.5% methionine, 3.90% methionine + cysteine, 2.2 Na, 2,100 kcal ME, 200,000 IU A, 40,000 IU D3, 500 mg E, 30 mg K3, 15 mg B1, 15 mg B2, 150 mg B3, 20 mg B6, 300 mg B12, 10 mg folic acid, 100 Mcg biotin, 1 mg Fe, 100 mg Cu, 1.2 Mn, 800 mg Zn, 15 mg I, 2 mg Se, 6 mg Co, 900 mg antioxidant (BHT). #NRC20 | |
The study involved 300 one day-old, unsexed Ross 308 broiler chicks. The birds were randomly divided into four treatment groups with 75 birds per treatment and 25 birds per replicate.
The chicks were housed in a hall that was divided into 12, 2×2 m pens where the floors were covered with a 5 cm layer of sawdust. A continuous lighting system (23 h of light and one hour of darkness) was used.
The chicks were given two diets ad libitum, starter and finisher, which contained 3,027 and 3,195.3 kcal kg–1 metabolic energy and 23 and 20% crude protein19, respectively (Table 1).
Threonine was added to the diet at 0, 300, 600 and 900 mg kg–1 to yield Th0, Th300, Th600 and Th900 treatments, respectively.
Productivity traits, including BW, WG, FI and FC, were measured weekly and at the end of the experimental period (35 days). At the end of the experimental period, 12 birds (6 males and 6 females per treatment) were slaughtered, cleaned and weighed to determine the Carcass Weight (CW) and Dressing Percentage (DP). The carcasses were then cut to determine the relative weight of the main (breast, thigh) and secondary (back (Bap), wing (Wp) and neck (Np)) regions of the carcass as well as the relative weight of the heart (Hp), liver (Lp) and spleen (SP). The weight of the bursa of Fabricius (BF) and amount of abdominal fat (Fp) were also measured.
Blood samples from 6 birds per replicate (3 males and 3 females) were collected on days 21 and 35. The blood samples were divided into tubes containing anticoagulants for blood tests to determine the Red Blood Cell (RBC) and White Blood Cell (WBC) counts, using a hemocytometer, (with an improved Neubauer ruling Chinese-made), H/L ratio20 and PCV%21. Hemoglobin (Hb) was estimated using the Darbkin method as described by Varley et al.22. The remaining blood samples were placed into 6 ml Gel tubes and centrifuged at 4,000 rpm for 10 min to separate serum from cellular components. The serum was stored at -20̊C prior to analysis. Glucose (Glu) concentrations were estimated according to a method described by Asatoor and King23. Total Protein (TP) concentrations were estimated according to the biuret method24 and the albumin concentration was estimated using a method described by Henry et al.25. Globulin levels were estimated based on a formula that involved the TP value and albumin levels26. Alkaline phosphatase (ALP) activity was determined using a method by Kind and King27, whereas aspartate transaminase (AST) and alanine transaminase (ALT) levels were estimated according to Reitman and Frankel28. Blood serum tests were performed using kits produced by Biomaghreb.
All statistical analyses were performed using the Statistical Package for Social Science (SPSS)29 version 21.0 for windows (SPSS Inc., Chicago, IL, USA) according to a Complete Randomized Design. Means were compared by Duncan’s Multiple Range Test30 with a significance level of 5%.
The addition of threonine at 600 and 900 mg kg–1 (Th600, Th900) resulted in significant increases (p<0.01) in BW of 14 day-old birds compared to birds that received 0 (Th0) or 300 mg kg–1 threonine (Th300) and this difference persisted through the end of the third week (21 days) (Table 2). By week four (28 days), the BW of birds receiving Th600 and Th900 differed significantly from those of Th0 and Th300, whereas birds in the Th900 group had higher BW than birds in the Th600 group (Table 2). By the end of the experimental period (35 days), Th900 birds showed a significant increase (p<0.05) in BW compared to birds receiving the other three treatments (Table 2).
During the first 7 days, there were no significant differences in Weight Gain (WG) among the threonine treatments (Th300, Th600 and Th900) compared with the control treatment (Table 3). However, during the days 8-14, diet supplemented with threonine significantly increased WG of birds of Th600 and Th900 groups compared to birds fed diet without threonine (Table 3). For the period between days 22-28 there was no significant difference between the Th0 and Th300 groups but significant increase in weight (p<0.01) was observed in Th600 and Th900 birds compared to Th0 birds.
| Table 2: | Effect of threonine supplementation on broiler live body weight (g bird–1) (Mean±SE) |
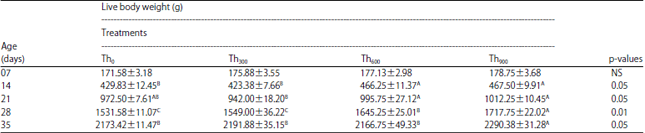 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A-CMeans within a row lacking a common superscript differ significantly | |
| Table 3: | Effect of threonine supplementation on body weight gain of broiler chickens (g bird–1) (Mean±SE) |
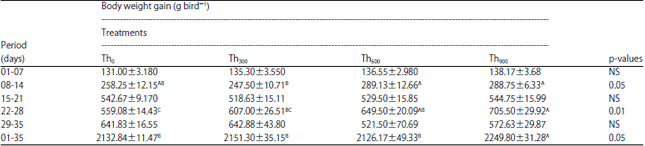 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A-CMeans within a row lacking a common superscript differ significantly | |
| Table 4: | Effect of threonine supplementation on broiler feed intake (g/bird/week) (Mean±SE) |
 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A-CMeans within a row lacking a common superscript differ significantly | |
| Table 5: | Effect of threonine supplementation on feed conversion ratio of broiler chickens (g feed/g gain) (Mean±SE) |
 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly | |
| Table 6: | Effect of threonine supplementation on carcass weight, dressing percentage and relative weight of heart, liver and gizzard (%) of broiler chickens (Mean±SE) |
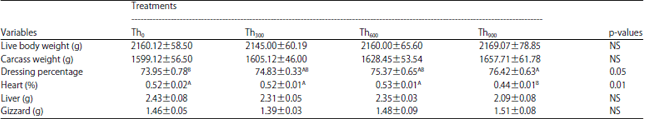 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly | |
The Th900 group had the highest WG across the entire experimental period (1-35 days) compared to the other three groups (Table 3).
Meanwhile, during the days 22-28, Feed Intake (FI) was increased significant (p<0.05) in the Th600 compared to Th0 groups but there was no significant difference between Th300 and Th900 treatment groups (Table 4). There were also no significant differences between treatments Th600, Th900 and Th0 during the days 29-35 but there was a significant decrease (p<0.05) in FI for birds in the Th300 group compared with those in the Th0 and Th900 groups. Over the entire experimental period (1-35 days), the FI was significantly higher in the Th600 group (p<0.01) compared to the Th0 and Th300 groups.
The Th900 group had the lowest feed conversion ratio (FC) compared to the Th0, Th300 and Th600 groups between the days 22-28 (p<0.05, Table 5). Between days 29-35, there were no significant differences in FC for any of the threonine treatment groups compared to Th0, although FC decreased significantly in the Th300 group (p<0.05) compared to that of the Th600 group. With respect to the overall experimental period (1-35 days), there were no significant differences between Th300, Th600, Th900 and Th0 in FC, although FC was significantly decreased (p<0.05) in the Th300 and Th900 groups compared to the Th600 group (Table 5).
Threonine supplementation did not affect the BW or Carcass Weight (CW) as well as the relative weights of the liver (LP) and gizzard (GP) across the treatment groups (Table 6).
| Table 7: | Effect of threonine supplementation on the relative weight of breast, thigh, back, wings and neck (%) in broiler chickens (Mean±SE) |
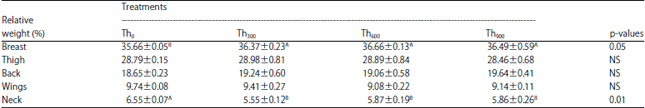 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly | |
| Table 8: | Effect of threonine supplementation on blood values for broiler chickens (Mean±SE) |
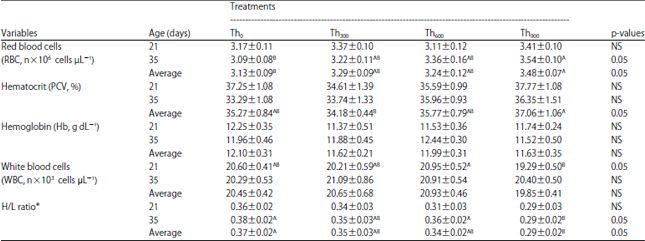 | |
Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly, *Heterophil/lymphocyte ratio | |
However, the DP was significantly increased in birds from the Th900 group (p<0.05) compared to the Th0 group. This change was accompanied by a significant decrease (p<0.05) in the relative weight of the heart (HP) compared to the Th0, Th300 and Th600 groups (Table 6).
Threonine supplementation significantly improved the relative breast weight (BRp) in the Th300, Th600 and Th900 groups compared to the Th0 group (Table 7). On the other hand, the Th300, Th600 and Th900 treatments had a significant decrease (p<0.01) in the neck weight (Np) compared to the control group. There were no significant differences among the threonine supplemented groups in weights of the wing (Wp), back (Bap) and thigh (THp) (Table 7).
Analysis of blood samples showed a significant increase (p<0.05) in the average number of RBCs on day 35 for the Th900 group compared to the other groups (Table 8). There were no differences in PCV% among the different treatment groups compared to the control on days 21 and 35 but the Th900 group showed significant increase (p<0.05) compare to the Th300 group in terms of the averages of the blood parameters assessed.
Despite changes in RBCs, threonine supplementation had no effect on Hb levels on the days assessed (Table 8).
Threonine treatments (Th300, Th600 and Th900) also did not significantly affect WBC levels. The Th900 group showed a significant decrease (p<0.05) in WBCs compared to the Th600 group on day 21 but there were no significant differences between Th0 and the experimental treatment groups (Th300, Th600 and Th900) on day 35 or on average (Table 8).
The Th900 group showed a significant decrease (p<0.05) in the heterophil/lymphocyte (H/L) ratio, which is a measure of long-term stress in chickens, on day 35 and on average compared to the control group. However, the H/L ratio was not significantly different among the three treatment groups.
There were no significant differences among the threonine treatment groups for serum glucose (Glu) on day 21, day 35 or on average compared with the control group (Table 9).
The Th300, Th600 and Th900 groups had higher total serum protein (TP) (p<0.05) compared with the Th0 group on day 21 and on average, although values on day 35 were similar, with the exception of the Th900 group (p<0.05).
| Table 9: | Effect of threonine supplementation on blood glucose, protein, calcium and phosphorous levels in broiler chickens (Mean±SE) |
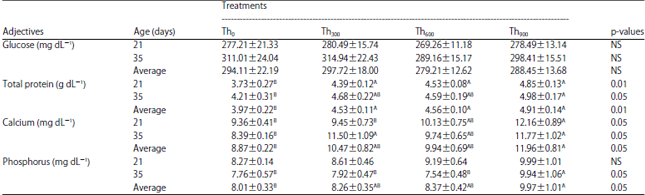 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly | |
| Table 10: | Effect of threonine supplementation on liver enzyme activity in blood serum from broiler chickens (Mean±SE) |
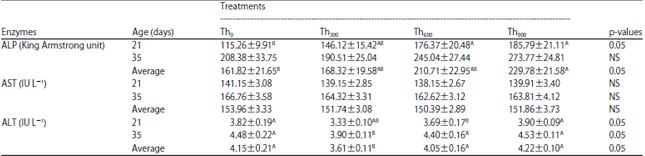 | |
| Th0, Th300, Th600 and Th900: Threonine supplementation to 0, 300, 600 and 900 mg kg–1, respectively. NS: Non-significant, A,BMeans within a row lacking a common superscript differ significantly | |
There was also a significant increase (p<0.05) in serum calcium (Ca) for the Th900 group compared with the control on days 21 and 35. The Th300 group had significantly improved Ca levels (p<0.05) compared to the Th0 group on day 35 but on day 21 and on average, the Th600 and Th0 groups were similar (Table 9). In addition, the Th300, Th600 and Th900 groups all had significantly higher (p<0.05) phosphorous (P) levels compared to the Th0 group on day 35.
Broiler chickens in the Th600 and Th900 groups showed significant increases (p<0.05) in ALP activity at day 35 but there were no significant differences (p<0.05) between the Th300 and Th0 groups. However, the Th900 group showed significant increase in ALP activity (p<0.05) (Table 10). In contrast, there were no significant differences in AST activity among the four groups (Table 10).
For ALT activity, the Th600 group showed a significant decrease (p<0.05) compared to the Th0 and Th900 groups but no significant differences were observed in Th300 group on day 21, whereas the value of ALT in Th900 group was higher than the other groups (Table 10). On day 35 and for the overall average, the Th300 group had a significant decrease (p<0.05) in ALT levels compared to the Th0, Th600 and Th900 groups.
Threonine supplementation did not affect Growth Hormone (GH) levels on day 21 (Fig. 1). Moreover, the Th300 and Th900 groups showed significant improvements (p<0.01) in GH levels on day 35 compared to the Th0 and Th600 groups. In addition, the Th900 group had significantly higher average GH levels (p<0.05) compared to the Th0 and Th600 groups but there were no significant differences between the Th300 and Th0 groups (Fig. 1).
In this study, supplementation of broiler chicken diets with 900 mg kg–1 threonine produced significant improvements in BW and WG (Table 2 and 3), which underscores the important role of this amino acid in promoting growth and muscularity31. Earlier studies showed that increasing the amount of dietary threonine increases nitrogen retention and reduces nitrogen loss in chickens32,33. Mucous membranes line the majority of the digestive tract and mucous membrane function is dependent on threonine for modulating the speed at which food passes through the digestive tract for effective nutrient absorption34.
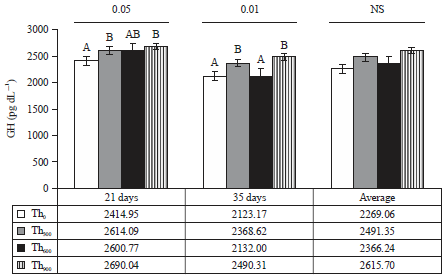 | |
| Fig. 1: | Effect of threonine supplementation on growth hormone concentration in blood from broiler chickens (Mean±SE) |
| Growth hormone values in blood samples were measured on 21 and 35 days and the average was taken across the experiment period | |
Moreover, threonine also maintain adequate secretion of digestive enzymes. Rezaeipour et al.35 demonstrated that increasing the amount of dietary threonine improved morphological traits of the small intestine in broiler chickens and these traits are reflected in an increased growth rate, whereas chickens fed diets lacking threonine had reduced nutrient absorption in the digestive tract.
Increased rates of nutrient absorption in the small intestine lead to increases in the availability of amino acids that can be used for protein synthesis, which is reflected in the increased levels of total protein (TP, Table 9) and activity of liver enzymes involved in metabolism36. These effects were associated with increased DP and relative breast weight and are reflected in the Th900 group (Table 6, 7).
The improvement in performance of birds fed the Th900 treatment highlight the role of threonine in increasing thyroid gland activity, including stimulation of hormone production. Azzam and El-Gogary37 and Wu38 reported that increasing the amount of dietary threonine led to increases in the concentrations of thyroxine (T4) and triiodothyronine (T3) hormones in chickens, which was likely due to the presence of threonine receptors in the thyroid gland important for the synthesis of T3. Thyroxine T4 is important for energy and protein metabolism39,40 and induces increased concentrations of the IGF-I hormone41. Appropriate amounts of T4 are needed for protein synthesis and maintenance of muscular mass as well as for enhanced function of GH42-44. The results of this study indicate that supplementation of diets with 600 or 900 mg kg–1 threonine increased GH levels (Fig. 1).
In addition to the pivotal roles of GH in muscularity and stimulation of T3, T4 and hormone IGF-I production36, GH also has roles in amino acid metabolism and protein synthesis that increase protein production45. Threonine supplementation may have improved feed palatability and this improvement is reflected by the increased FI for the Th600 and Th900 treatments (Table 4). The increased FI may also be related to hormonal control, since increased metabolic rates are associated with increased T4 concentrations. Moreover, GH levels can also affect the relationship between T4 and FI36,45.
The function of threonine in improving muscle growth and mass32 was evidenced by the significant improvement in DP (Table 6) and BRp (Table 7), which was likely manifested through increased absorption of digestive enzymes in the small intestine as well as increased availability of amino acids and rate of absorption of feed through the small intestine. This increase was associated with the increased concentration of protein in the blood (Table 9), T437 and GH (Fig. 1). Changes in these parameters are reflected in the increased body weight (Table 2) as well as DP (Table 6).
The improvement in productivity (Table 2, 3) and carcass traits (Table 6, 7) were associated with improvements in several physiological parameters, including RBCs and Hb (Table 8). The positive relationship of RBCs with BW and WG is reflected in increased metabolic rates manifested by T4 and GH levels43,46. T4 levels are also affected by erythropoietin, which increases productivity rates47 and simulates both RBCs and estrogen production48,49.
The significant decrease in the H/L ratio observed in this study is a strong indicator of increased amino acid levels50, whereas increases in the number of heterophils is an important indicator of immune system function51. Threonine increases globulin levels in the blood by increasing lymphocyte ratios14. The significant improvement in TP of birds that received threonine treatments (Table 9) is indicated by increased metabolic rates, especially if it is accompanied by body weight increases (Table 2) and high concentrations of Ca and P (Table 9). Threonine supplementation also increased ALP activity (Table 10), which is associated with an increased metabolic rate and muscle growth as well as increased GH levels (Fig. 1)49. Therefore, increased metabolic rates are reflected in increased ALP activity in the liver52.
The results of the present study reveal that diet supplemented with threonine can increase GH levels in the blood of the birds (Fig. 1). The GH mediates body and muscle growth through a variety of pathways, including stimulation of T4 and IGF-I production37.
CONCLUSION AND FUTURE RECOMMENDATION
Increased in GH levels improved growth and productive performance of broiler chickens. Use of threonine in broiler diets at a level of 900 mg kg–1 is recommended. Future studies should explore the effects of threonine supplementation on the expression of growth hormone genes.
This study found that threonine supplementation improves broiler production by increasing metabolic rates as well as total protein, calcium and phosphorus levels. It has been shown that threonine stimulates the pituitary gland to produce growth hormone, which contributes to an increase in thyroid activity and hormone production, resulting in improved broiler production. Threonine contributes to the maintenance of physiologic homeostasis, which is underscored by an improved H/L ratio. Increasing broiler production is important for improving the current economic situation in Iraq and developing countries as well as reducing the reliance on imported meat and eggs.