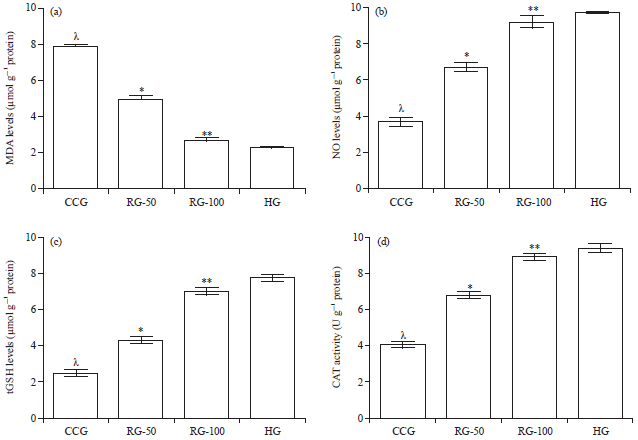
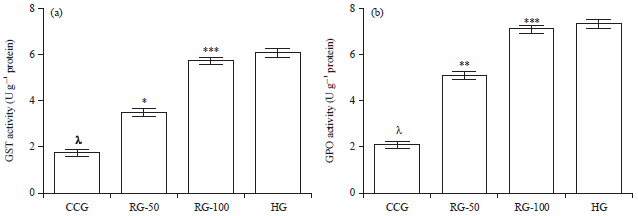
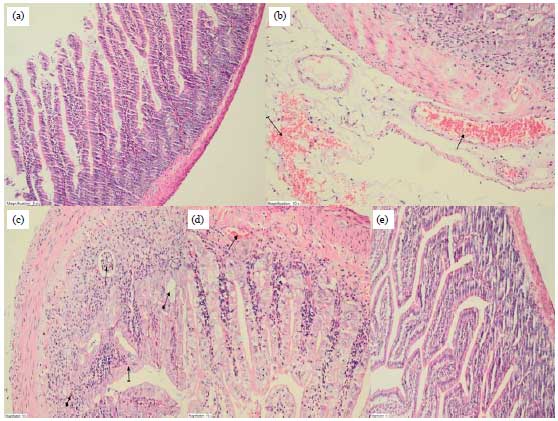
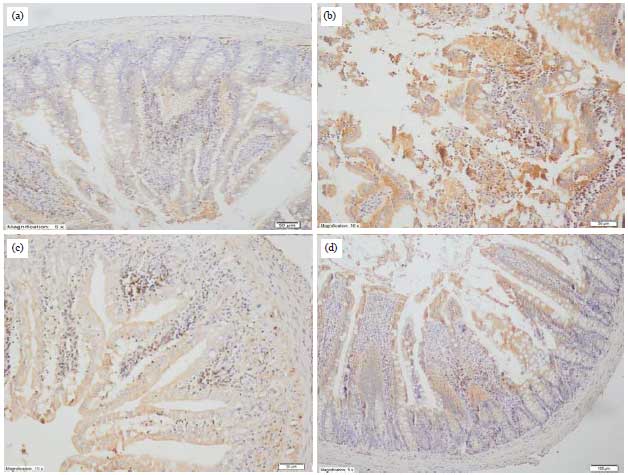
Research Article
Effect of Rutin on Cisplatin-induced Small Intestine (Jejunum) Damage in Rats
Department of Pediatrics, Neon Hospital, Erzincan, Turkey
Ulas Emre Akbulut
Department of Pediatric Gastroenterology, Training and Research Hospital, Antalya, Turkey
Orhan Cimen
Department of General Surgery, Faculty of Medicine, Erzincan University, Erzincan, Turkey
Abdulkerim Kolkiran
Department of Pediatrics, Faculty of Medicine, Erzincan University, Erzincan, Turkey
Selcuk Akturan
Department of Medical Education, Faculty of Medicine, Karadeniz Technical University, Trabzon, Turkey
Ferda Keskin Cimen
Department of Pathology, Faculty of Medicine, Erzincan University, Erzincan, Turkey
Murat Cankaya
Department of Biology, Faculty of Arts and Sciences, Erzincan University, Erzincan, Turkey
Arif Onur Eden
Department of Emergency, Faculty of Medicine, Erzincan University, Erzincan, Turkey
Zeynep Suleyman
Department of Nursing, Faculty of Health Sciences, Erzincan University, Erzincan, Turkey
Asli Ozbek Bilgin
Department of Pharmacology, Faculty of Medicine, Erzincan University, Erzincan, Turkey
LiveDNA: 90.23569