Review Article
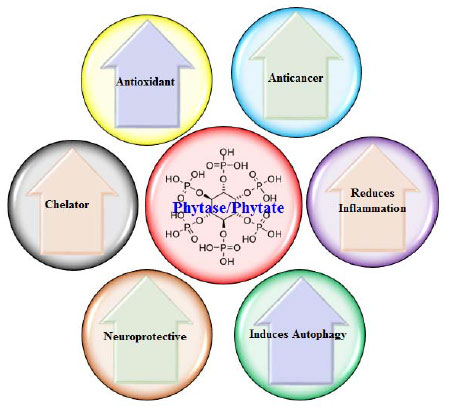
Anticancer and Nutraceutical Potentialities of Phytase/Phytate
Department of Biochemistry and Biotechnology, University of Gujrat, Pakistan
Muhammad Asgher
Department of Biochemistry, University of Agriculture Faisalabad, Pakistan
Khizar Hayat Bhatti
Department of Botany, University of Gujrat, Pakistan
Muddassar Zafar
Department of Biochemistry and Biotechnology, University of Gujrat, Pakistan
Zahid Anwar
Department of Biochemistry and Biotechnology, University of Gujrat, Pakistan