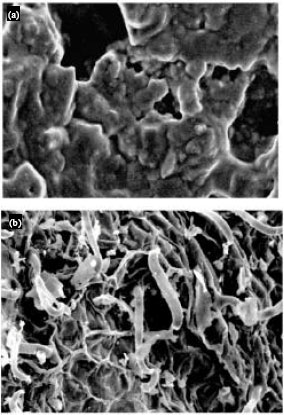
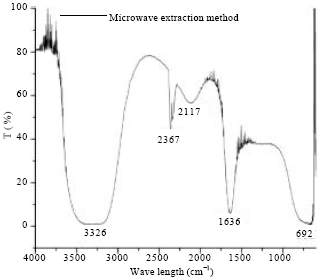
Research Article
Improved Extraction of Natural Blue dye from Butterfly Pea using Microwave Assisted Methodology to Reduce the Effect of Synthetic Blue Dye
Department of Biotechnology, National Institute of Technology, Durgapur-713209, India
Papita Das Saha
Department of Biotechnology, National Institute of Technology, Durgapur-713209, India
V. Ramya
Department of Biotechnology, National Institute of Technology, Durgapur-713209, India
Siddhartha Datta
Department of Chemical Engineering, Jadavpur University, Kolkata, India