Research Article
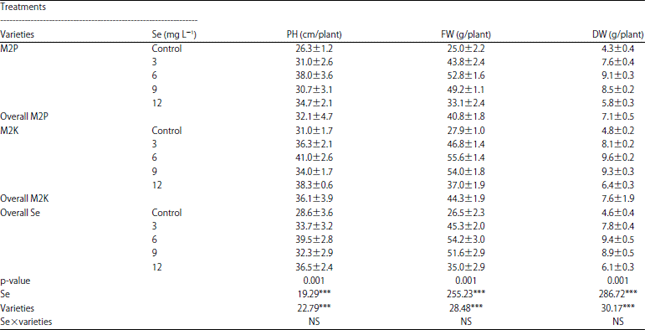
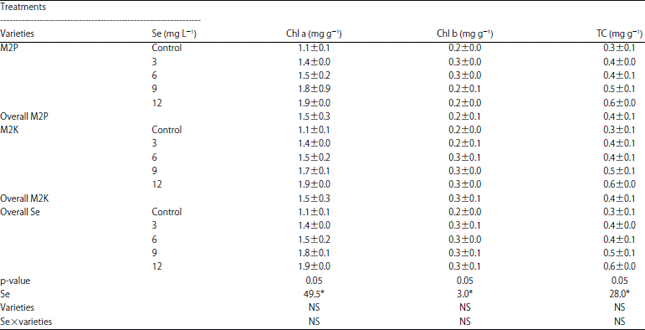
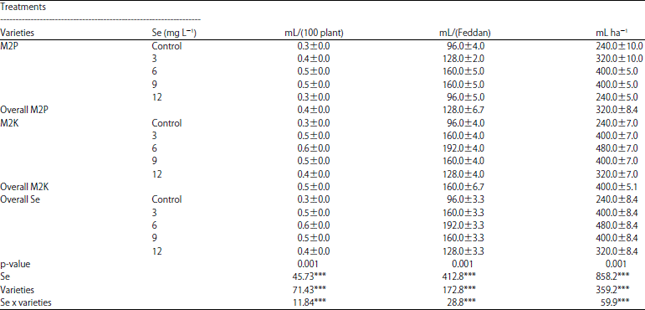
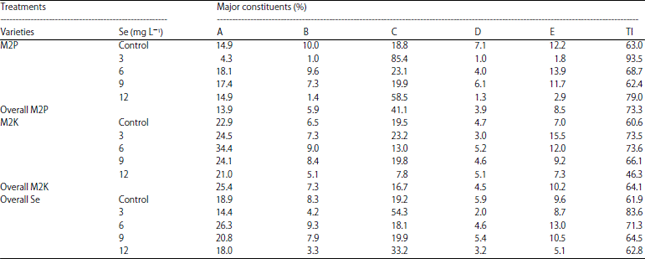
Selenium to Improve Growth Characters, Photosynthetic Pigments and Essential Oil Composition of Chives Varieties
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt
LiveDNA: 20.3160
Heba Mostafa Amer
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt
Hend Elsayed Wahba
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt
Saber Fayez Hendawy
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt
LiveDNA: 20.9481
Tamer Mohamed Abd El-Razik
Department of Medicinal and Aromatic Plants, National Research Centre, 12311 Dokki, Cairo, Egypt