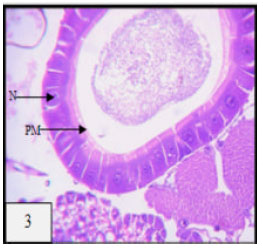
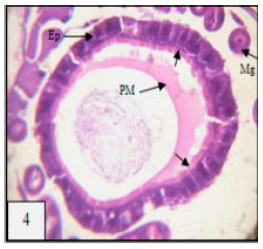
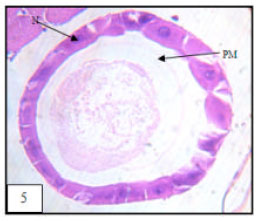
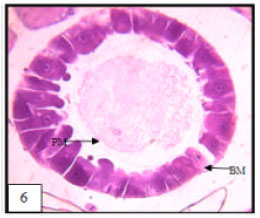
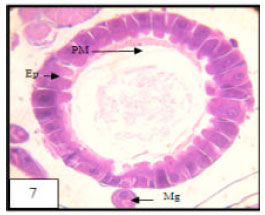
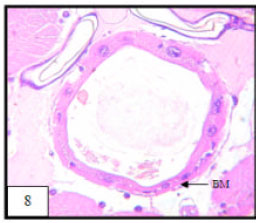
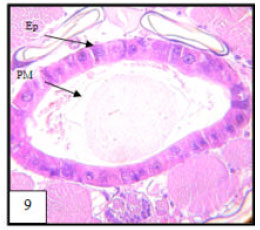
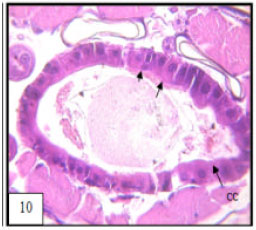
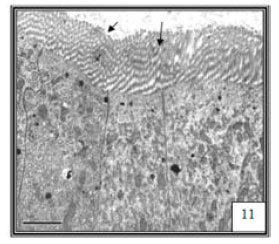
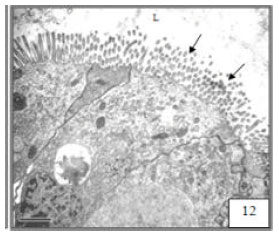
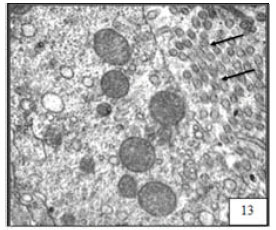
Research Article
Larvicidal, Histopathological and Ultra-structure Studies of Matricharia chamomella Extracts Against the Rift Valley Fever Mosquito Culex quinquefasciatus (Culicidae: Diptera)
Department of Zoology, Girls College, King Abdul-Aziz University, P.O. Box 19516, Jeddah 21445, Saudi Arabia