Research Article
Contribution of Rho/Rho-kinase Signalisation Pathway to Gentamicin Induced Nephrotoxicity in Mice
Department of Pharmacology, Faculty of Medicine, Cukurova University, 01330 Adana, Turkey
LiveDNA: 90.12212
Various studies showed that one of the main reasons for acute intrinsic renal insufficiency is aminoglycoside nephrotoxicity. Nephrotoxicity can be defined as nephrotoxic renal insufficiency. Kidneys are prone to be affected by toxic effects of the drugs and other endogenic and exogenic toxins due to their function in blood perfusion, metabolic activity and excretion. Renal tubular cells are exposed to high toxin concentrations more than other tissues because of their role in active tubular secretion, reabsorption and urine concentration mechanisms in kidneys. Due to this reason, renal tubules are direct targets of nephrotoxicity. Tubular necrosis is the significant feature of the nephrotoxicity. Because of this, it is also called acute tubular necrosis1.
Aminoglycoside antibiotics are widely used in control and treatment of the Gram (-) aerobic infections. Although, gentamicin has the most widespread application areas among these antibiotics, its usage is limited because of its nephrotoxic effects. Aminoglycoside antibiotics are responsible for nearly 10% of the acute renal insufficiency incidences. Due to this, aminoglycoside antibiotics are used as a model in acute renal insufficiency incidences1.
Rho GTPases comprise 1 of 5 distinct classes of Ras superfamily GTPase proteins. The canonical members of the Rho subfamily2 are RhoA, Rac1 and Cdc42. Rho GTPases function in a wide variety of cellular processes, including cell growth, motility, polarity and adhesion2.
Rho-kinase is a Ser/Thr protein kinase that was identified as a GTP-Rho-binding protein from bovine brain by affinity column chromatography on matrix-bound GTP-Rho. Rho-kinase was also identified as ROKα and ROCK2 ROKβ (also known as ROCK1) is an isoform of Rho-kinase (ROKα/ROCK2). Hereafter, both Rho-kinase/ROKα/ROCK2 and ROKβ/ROCK1 are referred to as Rho-kinase3,4. RhoA activates Rho-kinase then active Rho-kinase contributes intracellular processes.
Some studies showed that oxidative stress contributes gentamicin induced nephrotoxicity5. Oxidative stress increases intracellular Ca2+ levels6. Intracellular Ca2+ has relation with Rho/Rho-kinase signalization pathway3. Also, some physiologic conditions are same between gentamicin induced nephrotoxicity and diabetes and ischemia such as increasing oxidative stress7. Inhibition of Rho-kinase attenuates ischemia induced injury2. For this propose, here planed to investigate contribution of Rho/Rho-kinase signalization pathway on gentamicin induced nephrotoxicity.
Male mice (8 week old, balb/c, albino) that are obtained from the Experimental Animal Center in Çukurova University, in Adana are used in study. This study was approved by the Animal Care Committee and Ethics Committee of Cukurova University.
Mice are divided into two groups which are; control group and gentamicin group. To gentamicin group, 100 mg kg–1 gentamicin is applied intraperitoneally once a day for 9 days. Physiological serum is applied intraperitoneally to control group under same experimental conditions for 9 days. At the end of the protocol described above, cervical dislocation is applied to the mice. Kidneys of the mice are stored in eppendorf tubes at -80°C for later use in quantitative analysis.
Quantitative analysis
Tissue homogenization: Frozen tissue samples that are stored in Eppendorf tubes are treated with 3 mL g–1 RIPA (radio-immunoprecipitation assay) buffer, 30 μL PMSF (phenylmethanesulfonyl fluoride), 30 μL sodyum vanadate and 30 μL protease inhibitor. Then, homogenates are obtained by using ultrosonication on those tubes on ice. Homogenates are then centrifuged at 10.000 RPM for 10 min and supernatants are taken and pellets are discarded.
Protein quantification: Bradford method is used to quantify the protein in homogenized tissues. By using Bovine serum albumin (1 μg mL–1), 1, 2, 3, 5, 7, 8, 10 μg mL–1 standarts are prepared. Then, 10 μL is taken from every sample and completed to 100 μL by adding distilled water. Lastly, 1 mL Bradford solution is added to standards and samples, vortexed and absorbances at 595 nanometer are measured manually. Protein quantification (μg μL–1) is done according to the standart curve drawn in Prism software.
ELISA (enzyme linked immunosorbent assay) test: The ELISA test is used to examine the expression of RhoA, Rho-kinase II and to examine the activity of Rho-kinase enzymes.
Statistic analysis: Results were expressed as Means±SEM and n refers to the number of animals used for each experiments. Differences in results between tissues were tested by unpaired Student’s t-test. The p-values less than 0.05 were considered to be significant.
Quantification of concentration of RhoA enzyme by ELISA: Gentamicin treatment caused an increase in the RhoA protein expression in kidneys (Fig. 1). Mean values of RhoA concentrations for control and gentamicin groups are found to be 1173 pg mL–1 (SEM 235.0) and 1803 pg mL–1 (SEM 125.8), respectively.
Quantification of concentration of Rho-kinase II enzyme by ELISA: Gentamicin treatment caused an increase in the Rho-kinase II protein expression in kidneys (Fig. 2). Mean values of Rho-kinase II concentrations for control and gentamicin groups are found to be 32870 pg mL–1 (SEM 1454) and 45970 pg mL–1 (SEM 346.0), respectively.
 | |
| Fig. 1: | Effect of gentamicin treatment on RhoA expression. The data is presented as Mean±SEM. Differences between parameters of control and gentamicin treated group were analyzed by applying unpaired Student’s t-test. *Difference between control and gentamicin treated group is significant with p<0.05 |
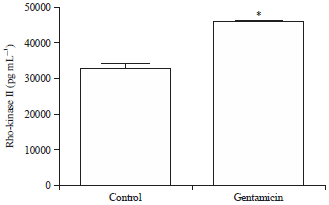 | |
| Fig. 2: | Effect of gentamicin treatment on Rho-kinase II expression. The data is presented as Mean±SEM. Differences between parameters of control and gentamicin treated group were analyzed by applying unpaired Student’s t-test. *Difference between control and gentamicin treated group is significant with p<0.05 |
Quantification of activity of Rho-kinase enzymes by ELISA: Gentamicin treatment caused an increase in the Rho-kinase activity in kidneys (Fig. 3). Mean values of Rho-kinase II activity for control and gentamicin groups are found to be 9.474 p-MYPT pg mg–1 (SEM 1.171) and 15.95 p-MYPT pg mg–1 (SEM 1.527), respectively.
The findings showed that gentamicin treatment activates Rho/Rho-kinase signalization pathway. Oxidative stress contributes gentamicin induced nephrotoxicity and also increases intracellular Ca2+ levels5,6. Intracellular Ca2+ is thought be responsible for activating Rho/Rho-kinase signalization pathway8.
A study showed that the oxidants activate RhoA9. While the guanine nucleotide binding properties of RhoA can be modulated directly by oxidants in vitro, few cellular studies have been conducted to determine the role of direct oxidation in regulating their activity9. Nevertheless, increasing evidence has tied RhoA to regulation by oxidation in cells. Given the relative reactivity of free radical oxidants compared with 2-electron oxidation, it is likely that free radical oxidation of reactive cysteines in the p-loop of Rho GTPases regulates Rho GTPase activity and signaling. Under reducing conditions, it is speculated that the free radical oxidants promote RhoA activation by facilitating nucleotide dissociation and GTP-loading, similar to the function of Guanin Exchange Factors (GEFs)10. However, under conditions of oxidative stress, these GTPases, which inactivates RhoA, may become inactivated2.
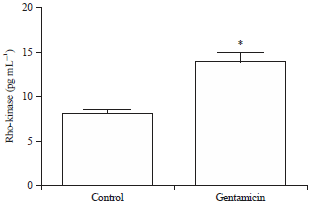 | |
| Fig. 3: | Effect of chronic gentamicin treatment on Rho-kinase activity. The data is presented as Mean±SEM. Differences between parameters of control and gentamicin treated group were analyzed by applying unpaired Student’s t-test. *Difference between control and gentamicin treated group is significant with p<0.05 |
On the other hand, findings of this study also showed that gentamicin treatment increased the activity and expression of Rho-kinase. Its increasing activity may be due to the increasing activity of RhoA. In addition, some studies reported that the expression and function of Rho-kinase is upregulated by inflammatory stimuli (e.g., angiotensin II and IL-1β) in hcVSMC with an involvement of PKC and NF-kappaB11. Moreover, various studies showed that the cyclooxygenase pathway, which functions in inflammation, accompanies gentamicin nephrotoxicity. In a previous study, selective cyclooxygenase-2 inhibitor is reported to be decreasing the nephrotoxicity caused by gentamicin12. Furthermore, gentamicin application is determined to be causing an increase in the activity of phospholipase13 A2. In another study, nephorotoxicity formed by gentamicin application caused an increase in iNOS enzyme14.
Findings of this study suggest that there is a strong correlation between Rho/Rho-kinase pathway and inflamation. Nowadays, use of gentamicin is low priority. It is chosen to create animal nephrotoxicity model. In conclusion, this study shows that gentamicin treatment enhanced Rho/Rho-kinase signalization pathway and helps us to understand the molecular mechanism of nephrotoxicity.
This study was funded by Cukurova University (TSA-2015-5198).