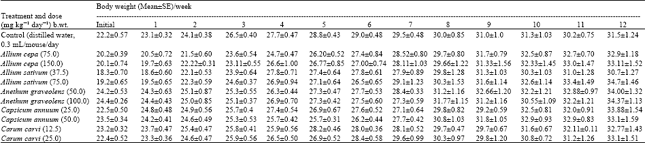
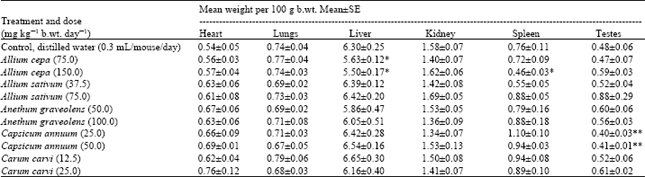
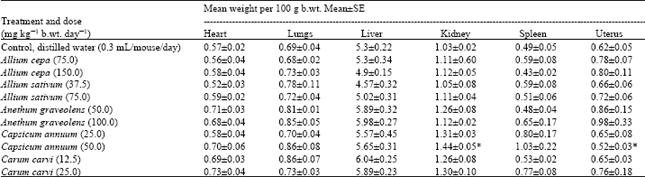
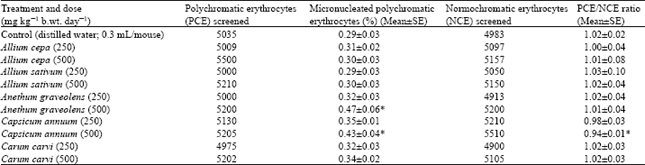
Research Article
Effect of Acute and Chronic Treatment of Common Spices in Swiss Albino Mice: A Safety Assessment Study
Department of Pharmacognosy and Medicinal, Aromatic and Poisonous Plants Research Center, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh-11451, Kingdom of Saudi Arabia
Tajdar Husain Khan
Department of Pharmacology, College of Pharmacy, Salman Bin Abdulaziz University, P.O. Box 173, Al-Kharj-11942, Kingdom of Saudi Arabia
Mohammed Al-Yahya
Department of Pharmacognosy and Medicinal, Aromatic and Poisonous Plants Research Center, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh-11451, Kingdom of Saudi Arabia
Ibrahim Al-Mofleh
Gastroenterology Unit (59), College of Medicine, King Khalid University Hospital, P.O. Box 2925, Riyadh-11461, Kingdom of Saudi Arabia
Syed Rafatullah
Department of Pharmacognosy and Medicinal, Aromatic and Poisonous Plants Research Center, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh-11451, Kingdom of Saudi Arabia