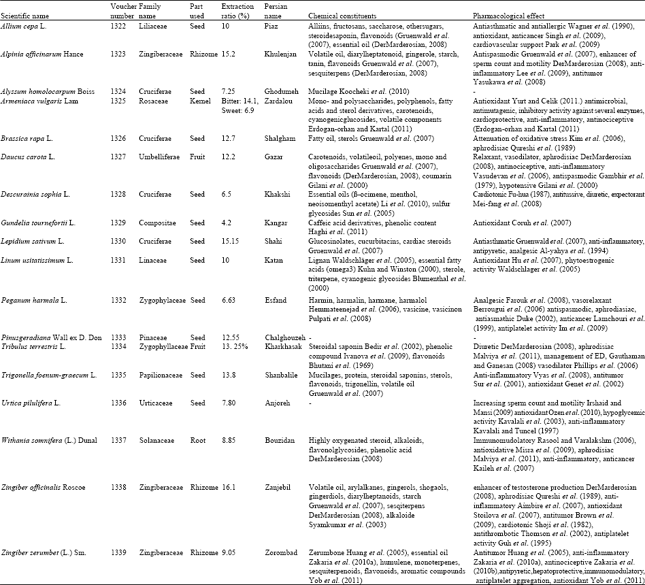
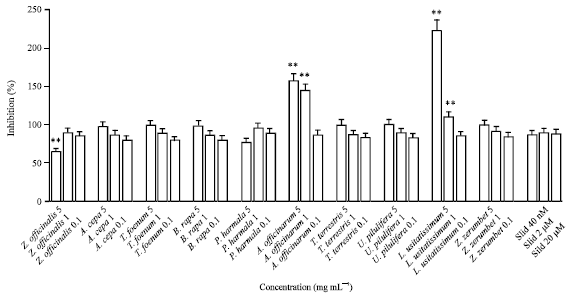
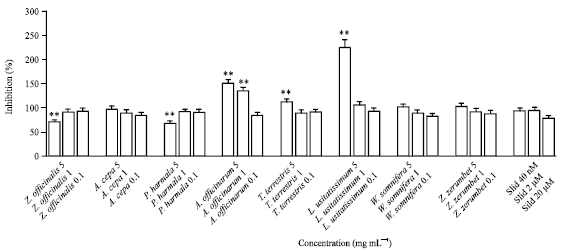
Research Article
Specifying Human Platelet cAMP and cGMP Phosphodiesterase Inhibitory Activity of the Plants Used in Traditional Iranian Medicine for the Purpose of Erectile Dysfunction
Department of Pharmacognosy, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
H. Azimi
Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
S. Ghiasi
Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
S. Hassani
Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, Iran
R. Rahimi
Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, Iran
S. Nikfar
Department of Pharmacoeconomics and Pharmaceutical Administration, Faculty of Pharmacy, Tehran University of Medical Sciences, Iran
Y. Ajani
Institute of Medicinal Plants (ACECR), Karaj, Iran
M.R. Shams-Ardekani
Department of Pharmacognosy, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
M. Abdollahi
Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences, Tehran, Iran