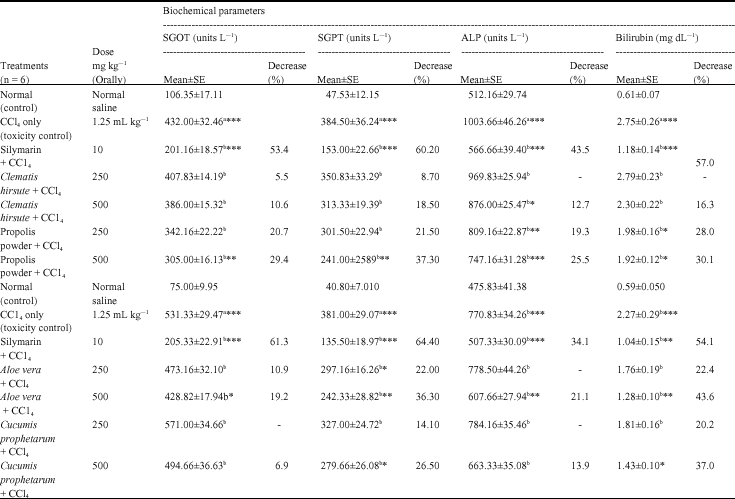
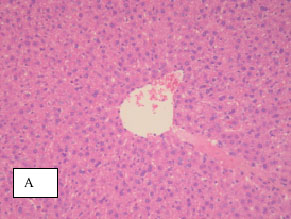
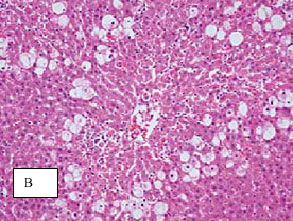
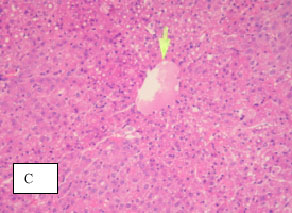
Research Article
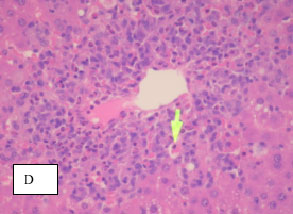
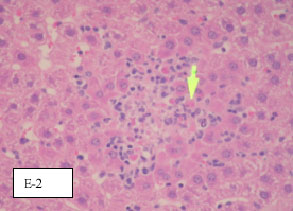
Evaluation of the Hepatoprotective Effect of Aloe vera, Clematis hirsute, Cucumis prophetarum and Bee Propolis Against Experimentally Induced Liver Injury in Rats
Department of Pharmacognosy, College of Pharmacy, King Saud University,P.O. Box 2457, Riyadh 11451, Kingdom of Saudi Arabia
Tawfik A. Al-Howiriny
Department of Pharmacognosy, College of Pharmacy, King Saud University,P.O. Box 2457, Riyadh 11451, Kingdom of Saudi Arabia
Maged S. Abdel-Kader
Department of Pharmacognosy, College of Pharmacy, King Saud University,P.O. Box 2457, Riyadh 11451, Kingdom of Saudi Arabia