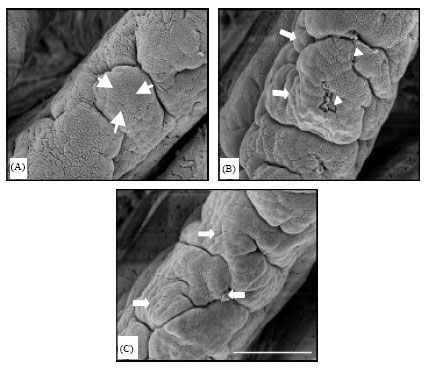
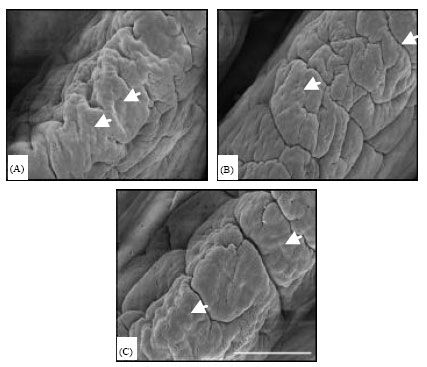
Research Article
Performance and Histological Changes of the Intestinal Villi in Chickens Fed Dietary Natural Zeolite Including Plant Extract
Laboratory of Animal Science, Faculty of Agriculture, Kagawa University, Miki-cho, Kagawa-ken, 761-0795, Japan
O. Khambualai
Laboratory of Animal Science, Faculty of Agriculture, Kagawa University, Miki-cho, Kagawa-ken, 761-0795, Japan
K. Yamauchi
Laboratory of Animal Science, Faculty of Agriculture, Kagawa University, Miki-cho, Kagawa-ken, 761-0795, Japan