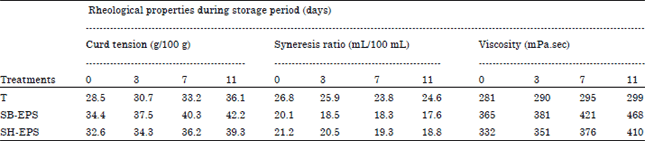
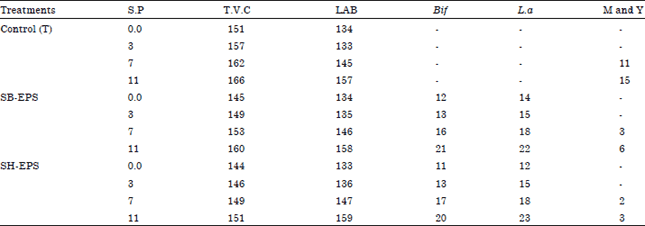
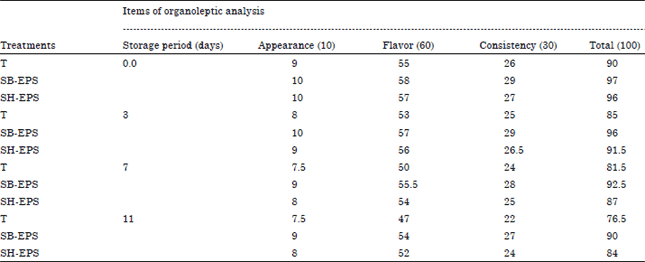
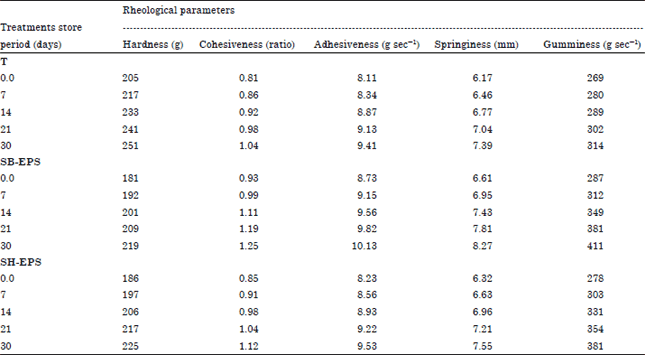
Research Article
Attributes of Low-Fat Yogurt and Kareish Cheese Made Using Exopolysaccharides Producing Lactic Acid Bacteria
Department of Dairy Research, Food Technology Research Institute, Agriculture Research Center, Ministry of Agriculture, Egypt
Amal Elsady Ibrahim
Department of Dairy Research, Food Technology Research Institute, Agriculture Research Center, Ministry of Agriculture, Egypt