Research Article
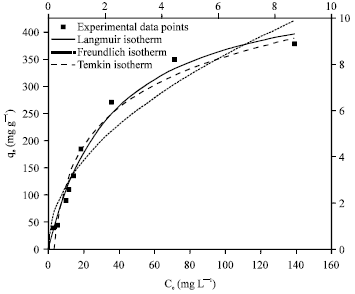
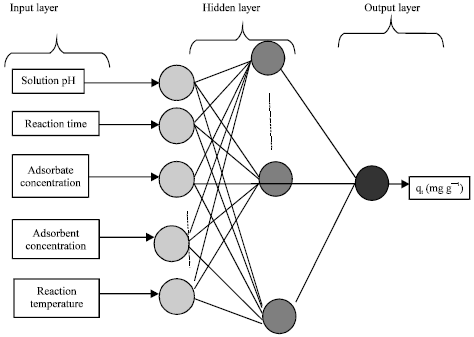
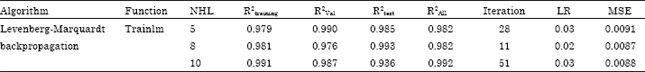
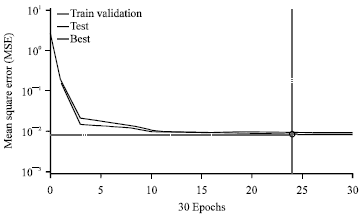
Prediction of Adsorption Capacity of Microwave Assisted Activated Carbon for the Decolorization of Direct Blue 86 by using Artificial Neural Network
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, Kharagpur-721302, India
Pinaki Ghosh
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, Kharagpur-721302, India
Jayanta K. Basu
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, Kharagpur-721302, India