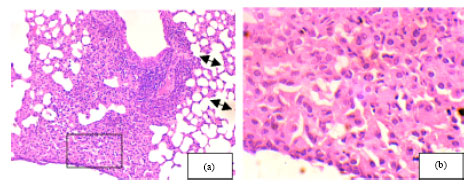
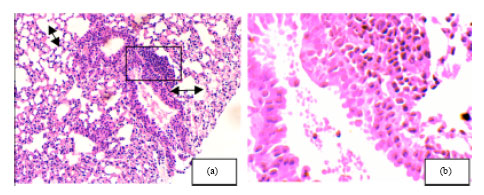
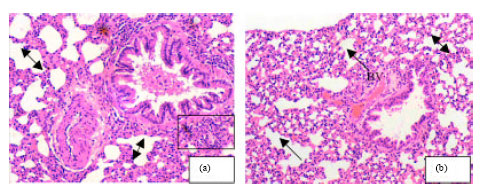
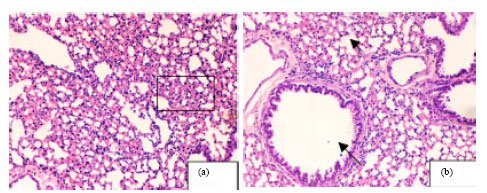
Research Article
Tumor Lung Cancer Model for Assessing Anti-neoplastic Effect of PMF in Rodents: Histopathological Study
Department of Biology, Faculty of Sciences, P.O. Box 80216, Jeddah 21589, Saudi Arabia
F. Khorshid
Department of Biology, Faculty of Sciences, P.O. Box 80216, Jeddah 21589, Saudi Arabia
H. Abu-araki
Animal House King Fahad Research Center, P.O. Box 80202, Jeddah, Saudi Arabia
A.M. Osman
Animal House King Fahad Research Center, P.O. Box 80202, Jeddah, Saudi Arabia