Research Article
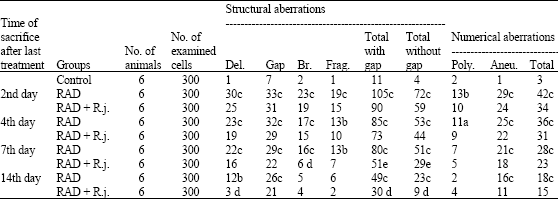
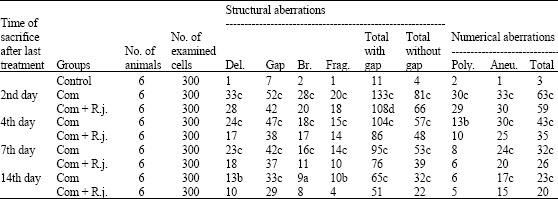
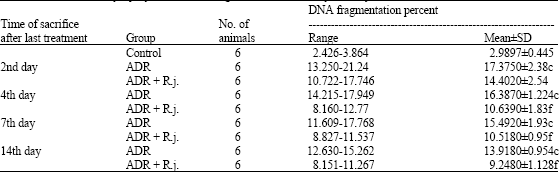
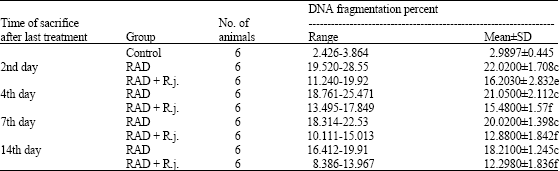
The Protective Role of Royal Jelly Against Mutagenic Effect of Adriamycin and Gamma Radiation Separately and in Combination
Department of Cell Biology, National Research Center, Dokki, Egypt
Othman E. Othman
Department of Cell Biology, National Research Center, Dokki, Egypt
Esraa A. Balabel
Department of Cell Biology, National Research Center, Dokki, Egypt
Soheir A. Abd-Elbaset
Department of Zoology, Faculty of Science, Al-Azhar University, Cairo, Egypt