ABSTRACT
The chloroform and methanol extract of the leaves Tithonia diversifolia and Jatropha gossypifolia were obtained using the cold extraction method. The anti-microbial activities of the extracts against ten human pathogens namely Staphylococcus aureus, Escherichia coli, Proteus mirabilis, Salmonella typhi, Bacillus subtilis, Shigella dysenteriae, Corynebacterium diptheriae, Pseudomonas aeroginosa, Streptococcus pneumoniae and Candida albicans were investigated using the agar well diffusion method. The results obtained showed that the chloroform and methanol extracts of the leaves of T. diversifolia and J. gossypifolia had antimicrobial effect against Salmonella typhi and Staphylococcus aureus with 12.0, 13.0, 12.0, 12.5, 20.0, 12.0, 6.0 and 18.0 mm as their zones of inhibition, respectively and Candida albicans was susceptible to all the extracts with zones of inhibition ranging from 17.0 to 35.0 mm. Other bacterial isolates were resistant except Pseudomonas aeroginosa that was susceptible only to Jatropha gossypifolia extracts. The anti-microbial activity competes well with standard antibiotics. Results of phytochemical screening showed the presence of saponin and tannin in the chloroform and methanol extracts of Tithonia diversifolia, phlobatannins and tannins in the methanol extract of Jatropha gossypifolia, phlobatannins and alkaloids in the chloroform extract of the same plant.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=tasr.2007.145.150
INTRODUCTION
Higher plants are sources of drugs, which have made important contribution to the welfare and quality of life of urban as well as rural communities especially in tropics and sub-tropics (Sofowora, 1993). During the early years of human existence, many plant materials by instinct, intuition of trial and error were used to combat different ailments. There has ever been an increasing interest investigating various plants for use in folkloric medicine.
There are many compounds in plants that have prominent effects on the animal system and some possess important therapeutic properties, which can be utilized in the treatment and care of human and other animal diseases. Thus the plant kingdom provides a tremendous reservoir of various chemical substances with potential therapeutic properties. There are several medicinal plants on earth that even the actual number is unknown. More medicinal plants were discovered at the end of 18th century while some of those that were previously discovered were eliminated due to high toxicity to mammals. The active ingredients of some medicinal plants were first extracted in 19th century, with series of pharmaceutical tests carried out on them to ensure their safety for making drugs.
In recent times, plant analyses have been effectively and efficiently carried out with the aid of advanced machines. This has yielded an ever-increasing number of medicinal constituent extract of plants. The use of plant extracts in the treatment of numerous human diseases has been reported by Khan et al. (2006). Many plants have been reported to exercise antimicrobial activities against various pathogens (Ahmed et al., 2006; Oladunmoye, 2006). Cragg and Newman (2005) also reported plants as sources of anti-cancer agents.
Jatropha gossypifolia is commonly known as Belly ache bush. The Yoruba speaking tribe of Nigeria call it Lapalapa. Jatropha gossypifolia is a bushy, gregarious shrub, up to 1.8 m 3-5 lobed, approximately 20 cm long and wide, with leaves having a long petiole, covered with glandular hairs. The seeds are greenish capsule-like seeds. The leaf stalks are covered with coarse dark brown hairs and the young leaves are sticky. It has thin, often greenish bark, which exudes copious amount of watery sap when cut. It is widely cultivated as ornamental. It prefers arid environment.
This plant originated from Brazil and it is now cultivated widely in tropical countries throughout the world. The root, stems, leaves, seeds and fruits of the plant have been widely used in traditional folk medicine in many parts of West Africa. The young stem of the plant is used as toothbrush as well as to clean the tongue in the treatment thrush. The tuber of the plant grinded into a paste is also locally used in the treatment of hemorrhoids. This plant is also traditionally planted along slopes in Nigeria for controlling water erosion.
Tithonia diversifolia is a stout shrubby herb, which grows to about 2-3 m (8-10 ft) in height. It is a perennial herb. The leaves are alternate with a blade having 3-5 lobes. The leaves are mostly sub-ovate, acute serrate and about 10-40 cm long. The flowers are borne on peduncle that is between 7-20 cm long, which is like daisies but bigger in size, about 6-8 cm in diameter. They are bright yellow in color. The plant is a native of Central America. The common names of Tithonia diversifolia are sunflower, tree marigold, Mexican sunflower, wild marigold and among others. It has been known to relieve rheumatism and the flowers are beneficial in treatment eye diseases (Stone, 1970). Active ingredients of medicinal plants are usually more concentrated in storage organs such as leaves, stems, bark and roots.
The leaf of the plant J. gossypifolia is known to be used in folk medicine to clean the tongue of thrush. This study is therefore to investigate the inhibitory potency of the leaf extract against Candida species with a view to recommending it for further purification and use in the production of drug in pharmaceutical industries to combat Candida infections. The use of other pathogens is to discover if it also possess antimicrobial activity against other pathogens. T. diversifolia, a plant used in folk medicine is been investigated to harness its inhibitory potentials against pathogens.
MATERIALS AND METHODS
Collection and Identification of Test Plants
The plant Jatropha gossypifolia (Belly ache bush) was collected at a garden along Atibiti layout, Ijapo Housing estate, Akure, Nigeria, while Tithonia diversifolia was collected at off campus, Federal University of Technology, Akure. The plants were authenticated at the Crop, Soil and Pest Department, Federal University of Technology, Akure, Nigeria.
Collection of Test Organisms
The test organisms included Staphylococcus aureus, Escherichia coli, Proteus mirabilis, Salmonella typhi, Bacillus subtilis, Shigella dysenteriae, Corynebacterium diptheriae, Pseudomonas aeruginosa, Streptococcus pneumoniae and Candida albicans, obtained from University College Hospital (UCH) Ibadan, Oyo-State, Nigeria.
Preparation of Extracts
Methanol Extract
The fresh leaves of Jatropha gossypifolia (J) and Tithonia diversifolia (T) were harvested and washed inside distilled water so as to remove dust and other foreign particles. The leaves were then left on a clean surface to air dry, until well-dried and grinded to fine powder using a blender. Exactly 450 g of the powdered leaf of Tithonia diversifolia and 300 g of Jatropha gossypifolia each measured separately into different containers and soaked with methanol. The mixtures were allowed to stand for 72 h and then filtered with muslin cloths. The filtrates were air-dried to obtain the leaf extracts, JM (Jatropha gossypifolia methanol extract) and TM (Tithonia diversifolia methanol extract), respectively.
Chloroform Extract
Exactly 450 g of Tithonia diversifolia and 300 g of Jatropha gossypifolia were soaked in chloroform following the procedure for methanol extraction. The subsequent extracts obtained from these were labeled TC (Tithonia diversifolia chloroform extract) and JC (Jathropha gossypifolia chloroform extract), respectively.
Antimicrobial Assay or Sensitivity Test
The pour plate method was adopted for this test. About 1 mL of Nutrient broth containing the test organism was introduced into a sterile petri dish and an already prepared sterile medium that has cooled to 45°C was poured on it. Nutrient agar medium was used for bacteria while potato dextrose agar medium was used to cultivate fungi. The plates were gently agitated for proper mixture and the agar was allowed to solidify. Afterwards, wells were dug in the plates with the aid of a sterile cork borer of 8 mm in diameter. With the aid of a sterile syringe, 25 mg mL-1 of the extracts were transferred into the wells made. About 0.4 mL of each extract was introduced into different wells. The plates were then incubated at 37°C for 24 h, after which they were observed for clear zones around the wells indicative of zones of inhibition, which were measured and recorded in mm. The Minimum Inhibitory Concentration (MIC) of the extracts was determined on the only susceptible organisms. This was investigated by varying the concentration of the extracts.
Antibiotics Sensitivity Test
Commercial antibiotic impregnated paper disc was placed on a seeded agar and incubated at 37°C for 24 h and the zone of inhibition was measured.
Phytochemical Screening
Test for Alkaloids
About 0.5 g of each plant extract was stirred with 5 mL of 1% aqueous hydrochloric acid on a steam bath. One milliliter of the filtrate was treated with a few drops of Dragendorff’s reagent. Turbidity or precipitation with this reagent indicated the presence of alkaloids in the extracts.
Test for Saponins
About 0.5 g of each plant extract was shaken with water in a test tube. Frothing which persisted on warming indicated presence of saponins.
Test for Phlobatannins
Aqueous sample of the plant extracts was boiled with 1% aqueous hydrochloric acid. Deposition of a red precipitate indicated the presence of phlobatannins.
Test for Tannins
About 5 g of each plant extracts was stirred with 10 mL of distilled water. It was filtered and ferric chloride reagent was added to the filtrate. A blue-green, green or blue-black precipitate indicated the presence of tannins.
This experiment was carried out July-August, 2006, at the Microbiology research laboratory, Federal University of Technology, Akure, Nigeria.
RESULTS AND DISCUSSION
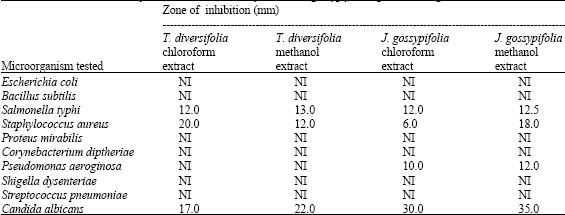
Results obtained from this research indicates that chloroform and methanol extracts of Tithonia diversifolia and Jatropha gossypifolia had inhibitory effect on Salmonella typhi and Staphylococcus aureus with zones of inhibition ranging from 6.00 to 18.00 mm (Table 1). The antimicrobial activity was more pronounced on the Gram positive bacteria (Staphylococcus aureus) than the Gram-negative bacteria (Salmonella typhi). The reason for the difference in sensitivity between Gram positive and the Gram negative bacteria may be ascribed to the differences in morphological constitutions between these microorganisms. Gram negative bacteria have an outer phospholipidic membrane carrying the structural lipopolysaccharide components. This makes the cell wall impermeable to antimicrobial chemical substances. The Gram positive bacteria on the other hand have only an outer peptidoglycan layer which is not an effective permeability barrier. Therefore, the cell walls of Gram negative organisms which are more complex than the gram-positive ones acts as a diffusional barrier and make them less susceptible to the antimicrobial agents than are Gram-positive bacteria (Nostro et al., 2000; Hodges, 2002). In fact, Gram negative bacteria are frequently reported to have developed multi-drug resistance to many of the antibiotics currently available in the market of which Escherichia coli is the most prominent (Alonso et al., 2000; Sader et al., 2002). Therefore, it is not surprising that Escherichia coli was the least responding bacterial strain to the test plant extracts. Despite this permeability differences, however, some of the extracts have still exerted some degree of inhibition against Gram negative organisms like Salmonella typhi and Pseudomonas aeruginosa hence the extract can be referred to as having a broad spectrum activity.
Although Bacillus subtilis is a Gram positive bacteria like Staphylococcus aureus, the test agents showed no potency on it (Table 1). The presumed reason for this resistance may be due to the presence of oval centrally located endospore, which serves as an encystment against the extracts.
Salmonella typhi and Pseudomonas aeruginosa were the only Gram negative bacteria that were inhibited by the test agents. Pseudomonas aeruginosa was however, resistant to the extracts of Tithonia diversifolia but it was susceptible to the extracts of Jatropha gossypifolia.
| Table 1: | Antimicrobial activity of extracts of T. diversifolia and J. gossypifolia against test organism |
 | |
| NI: No Inhibition | |
| Table 2: | The minimum inhibitory concentration of the extracts of T. diversifolia and J. gossypifolia |
 | |
| TC: T. diversifolia chloroform extract; TM: T. diversifolia methanol extract; JC: J. gossypifolia chloroform extract; JM: J. gossypifolia methanol extract; ND: Not Determined | |
| Table 3: | The phytochemical constituents of chloroform and methanol extracts of T. diversifolia and J. gossypifolia |
 | |
| + Present; - Absent; TM: T. diversifolia methanol extract; TC: T. diversifolia chloroform extract; JM: J. gossypifolia methanol extract; JC: J. gossypifolia chloroform extract | |
| Table 4: | Antibiotics sensitivity test |
 | |
| Am-Amoxyllin (25 μg); Ofl-Ofloxacin (25 μg); Str-Streptomycin (10 μg); Chl-Chloraphenicol (30 μg); Cef-Ceftriazone (30 μg); Gen-Gentamycin (10 μg); Pef-Pefloxacin (5 μg); Tet-Tetracycline (30 μg); Aug-Augmentin (30 μg); Cot-Cotrimazole (25 μg); I-Inactive | |
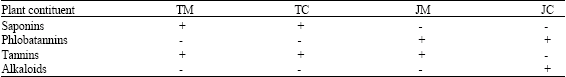
It is to be noted that phlobatannin was present in the chloroform and methanol extracts of Jatropha gossypifolia and conspicuously absent in the extracts of Tithonia diversifolia (Table 3). The presence or absence of this component may be an explainable reason for the difference in their ability to be able to inhibit or not inhibit Pseudomonas aeruginosa, because phytochemicals have been investigated and found responsible for various activities of plants such as cytoprotective, immunodulatory, immunoadjuvant and antimicrobial potential (Guatam et al., 2004a, b; Luc and Arnold, 2005; Abiy et al., 2005).
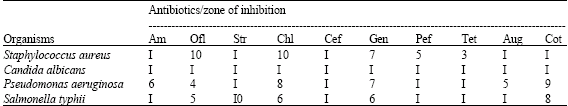
Fungi are mostly resistant to antimicrobial agents, even as shown in the antibiotic sensitivity test (Table 4). However, from this study, Candida albicans was inhibited by all the extracts of both plants, though the MIC of Jatropha gossypifolia chloroform extract on it was 25 mg mL-1 (Table 2). This explains that the plants can be used in the treatment of Candida infection.
Based on the initial antimicrobial screening test, Tithonia diversifolia and Jatropha gossypifolia were further studied for the determination of their Minimum Inhibitory Concentration (MIC).
The MIC value of TC, JC and JM against Salmonella typhi and Pseudomonas aeruginosa was 6.25 mg mL-1 while the value for TM against S. typhi was 3.125 mg mL-1 as shown on Table 2. The least MIC value recorded against Candida albicans exhibited by JM was 6.25 mg mL-1 and the highest value was 25 mg mL-1 by JC (Table 2).
From the results shown on Table 1, it was observed that the methanol extracts of both Tithonia diversifolia and Jatropha gossypifolia were more active against the test isolates than the chloroform extracts except in the case of Tithonia diversifolia against Staphylococcus aureus that was otherwise with 20 mm as zone of inhibition. A similar result was obtained for Vernonia tenoreana where the methanol extract was found to exercise higher antimicrobial activities against selected pathogens than the chloroform extracts of the same plant (Ogundare et al., 2006). Rios and Reccio (2005) also said that the solvent used and the extraction system may both modify the final results of an experiment, which also explains the reason for the higher potency of the methanol extracts.
The phtyochemical constituents (tannins and saponins) present in the methanol and chloroform extracts of Tithonia diversifolia are the same, although further advanced extractive systems can show certain phtyochemical(s) present in the extracts.
The result of the antimicrobial activity of the plant extracts competes well with the standard antibiotics (Table 4).
Although the extracts of these plants were able to inhibit the growth of certain bacteria and Candida albicans in vitro. However, an in vivo antimicrobial as well as the toxicological analysis of the plant extracts which were not carried out in this experiment need to be investigated to know the safety dose of the plants in case of oral administration to humans. The in vivo test will reveal whether or not the plants’ antimicrobial activity will reduce, increase, or give the same results as in the in vitro test.
REFERENCES
- Yenesew, A., S. Derese, J.O. Midiwo, C.C. Bii, M. Heydenreich and M.G. Peter, 2005. Antimicrobial flavonoids from the stem bark of Erythrina burttii. Fitoterapia, 76: 469-472.
CrossRefDirect Link - Ahmed, F., M.A. Islam and M.M. Rahman, 2006. Antimicrobial activity of Leonurus sibiricus aerial parts. Fitoterapia, 77: 316-317.
CrossRef - Alonso, R., A.F. Aranguiiz, K. Colom, A. Herreras and R. Cisterna, 2000. Profile of bacterial isolates and antimicrobial susceptibility. Revista Espanola de Quinioterapia, 13: 384-393.
PubMed - Cragg, G.M. and D.J. Newman, 2005. Plants as source of anti cancer agents. J. Ethnopharmacol., 100: 72-79.
PubMed - Guatam, M., S. Diwanay, S. Gairola, Y.S. Shinde, P. Patki and B. Patwardhan, 2004. Immunoadjuvant potential of Asparagus racemosus aqeous extract in experimental system. J. Ethnopharmacol., 91: 251-255.
PubMed - Gautam, M., S.S. Diwanay, S. Gairola, Y.S. Shinde, S.S. Jadhav and B.K. Patwardhan, 2004. Immune response modulation to DPT vaccine by aqueous extract of Withania somnifera in experimental system. Int. Immunopharmacol., 4: 841-849.
CrossRefDirect Link - Khan, M.R., A.D. Omoloso and Y. Barewai, 2006. Antimicrobial activity of Maniltoa schefferi extracts. Fitoterapia, 77: 324-326.
CrossRef - Nostro, A., M.P. Germano, V. D'Angelo, A. Marino and M.A. Cannatelli, 2000. Extraction methods and bioautography for evaluation of medicinal plant antimicrobial activity. Lett. Applied Microbial., 30: 379-384.
CrossRefPubMedDirect Link - Oladunmoye, M.K., 2006. Comparative evaluation of antimicrobial activities and phytochemical screening of two varieties of Acalypha wilkesiana. Trends Applied Sci. Res., 1: 538-541.
CrossRefDirect Link - Rios, J.L. and M.C. Recio, 2005. Medicinal plants and antimicrobial activity. J. Ethnopharmacol., 100: 80-84.
CrossRefPubMedDirect Link - Sofowora, A., 1993. Medicinal Plants and Traditional Medicine in Africa. 2nd Edn., Spectrum Books, Ibadan, Nigeria, ISBN-13: 9789782462190, Pages: 289.
Direct Link - Pieters, L. and A.J. Vlietinck, 2005. Bioguided isolation of pharmacologically active plant components, still a valuable strategy for the finding of new lead compounds? 100th volume special section: Perspectives of ethnopharmacology. J. Ethnopharmacol., 100: 57-60.
CrossRef - Pieters, L. and A.J. Vlietinck, 2005. Bioguided isolation of pharmacologically active plant components, still a valuable strategy for the finding of new lead compounds? 100th volume special section: Perspectives of ethnopharmacology. J. Ethnopharmacol., 100: 57-60.
CrossRef








