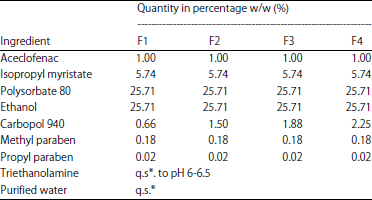
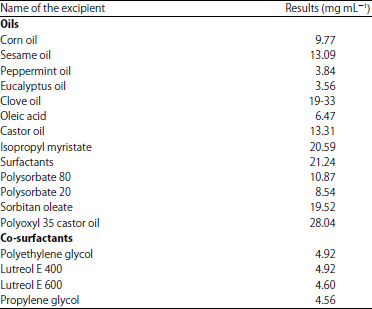
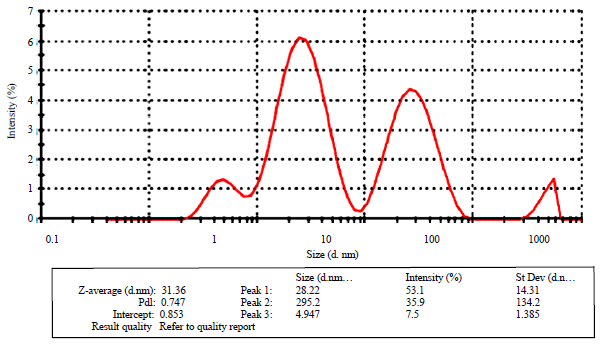
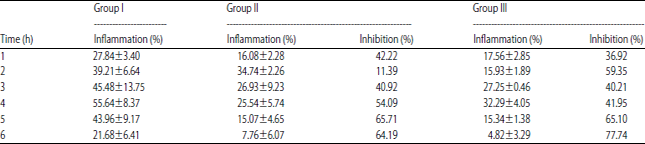
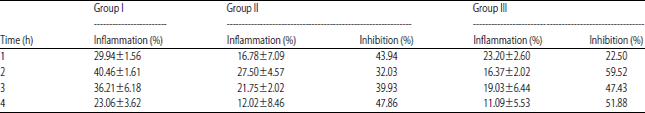
Research Article
Formulation, Development and Evaluation of a Novel Polymer-based Anti-Inflammatory Emulgel of Aceclofenac
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM�s NMIMS, Mumbai, Maharashtra
Mihir Invally
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM�s NMIMS, Mumbai, Maharashtra
Sarika Wairkar
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM�s NMIMS, Mumbai, Maharashtra
Sukhwinder Bhullar
Department of Mechanical Engineering, University of Victoria, British Columbia, Canada
Ginpreet Kaur
Shobhaben Pratapbhai Patel School of Pharmacy and Technology Management, SVKM�s NMIMS, Mumbai, Maharashtra
LiveDNA: 91.3069