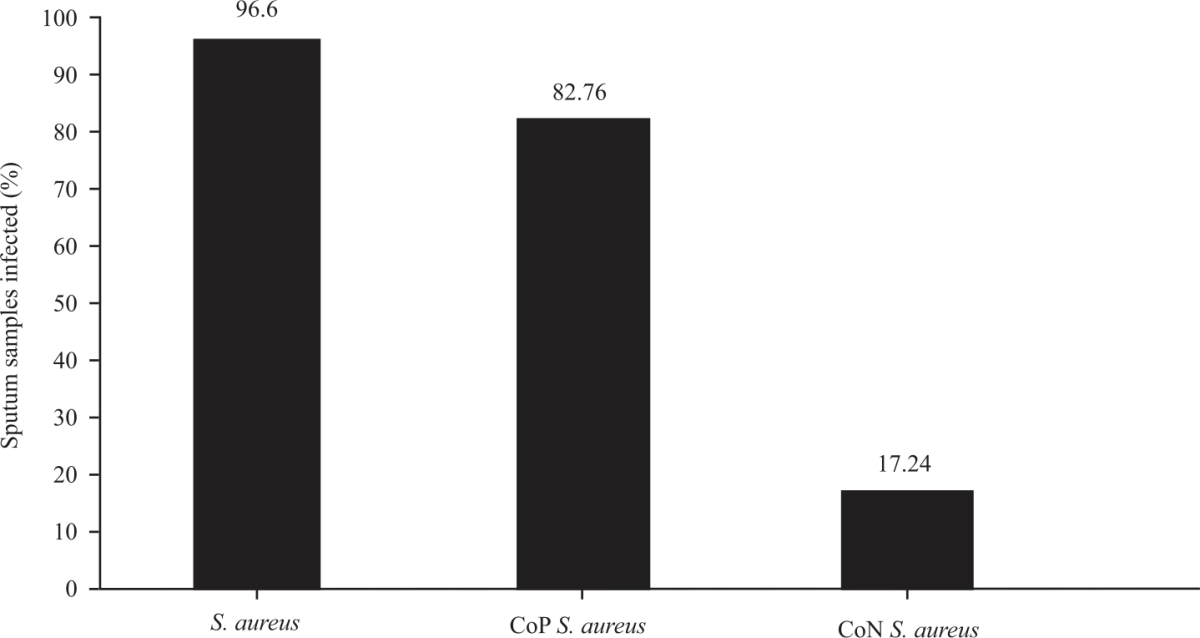
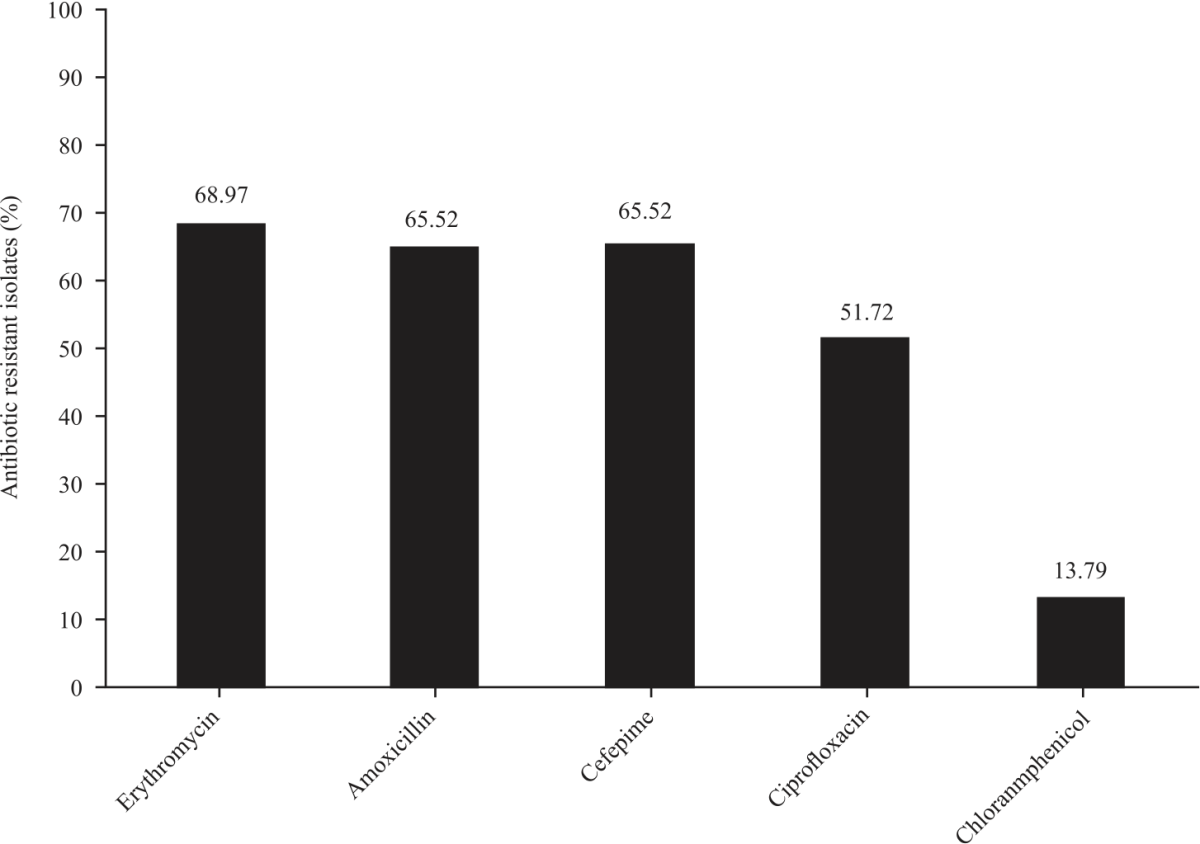
Research Article
Activity of Terminalia chebula and Erythrina variegata Extracts Against Pathogen Causing Respiratory Tract Infection
Department of Genetic Engineering and Biotechnology, University of Chittagong, Chattogram-4331, Bangladesh
Dil Umme Salma Chowdhury
Department of Genetic Engineering and Biotechnology, University of Chittagong, Chattogram-4331, Bangladesh
Abdul Mannan
Department of Chemistry, University of Chittagong, Chattogram-4331, Bangladesh
Nazneen Naher Islam
Department of Genetic Engineering and Biotechnology, University of Chittagong, Chattogram-4331, Bangladesh
LiveDNA: 880.21410