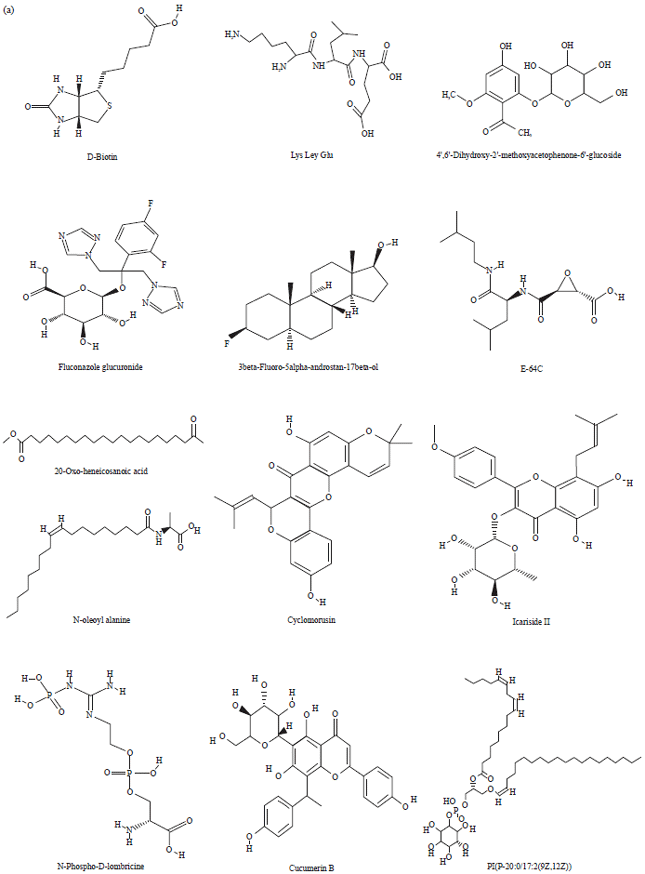
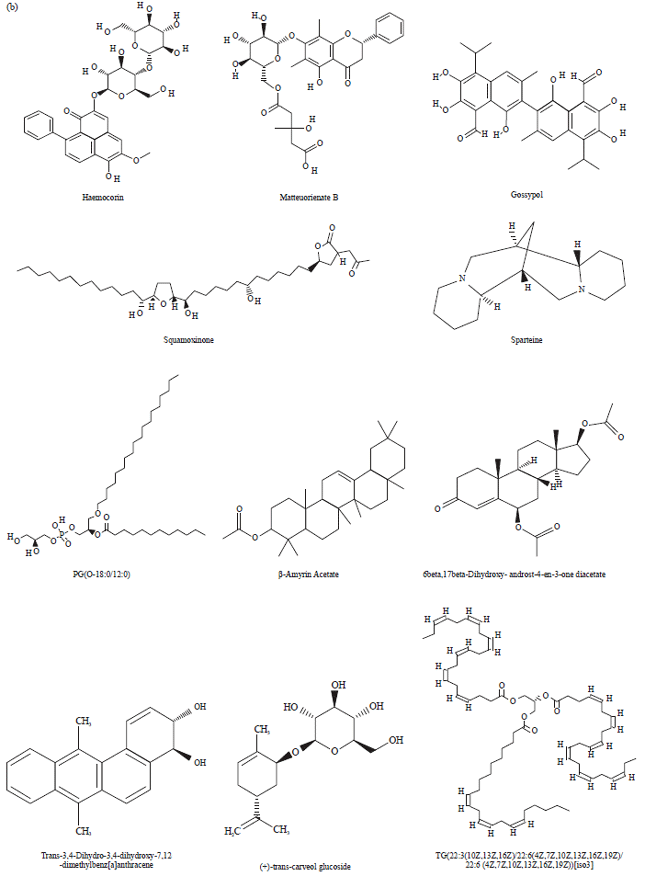
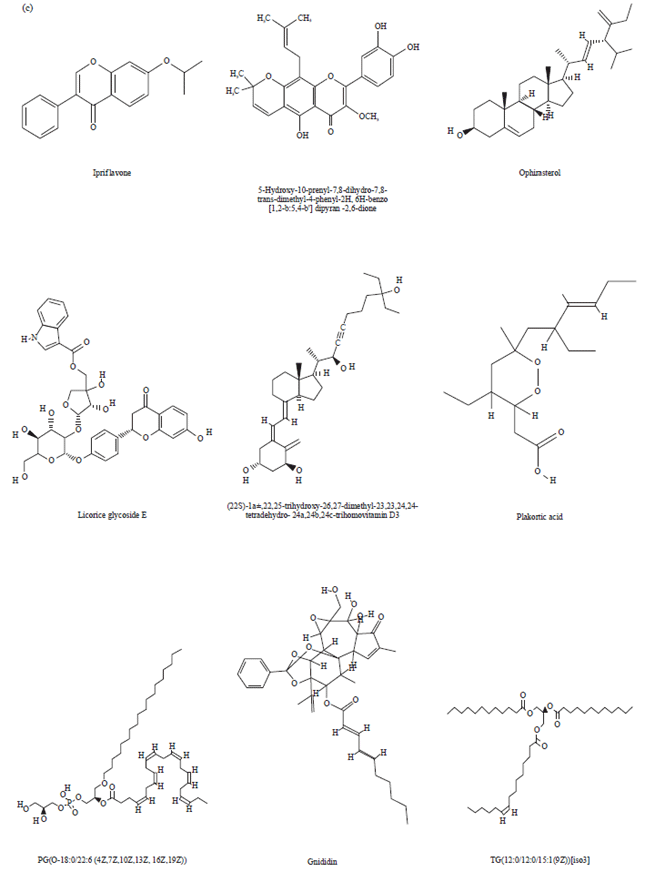
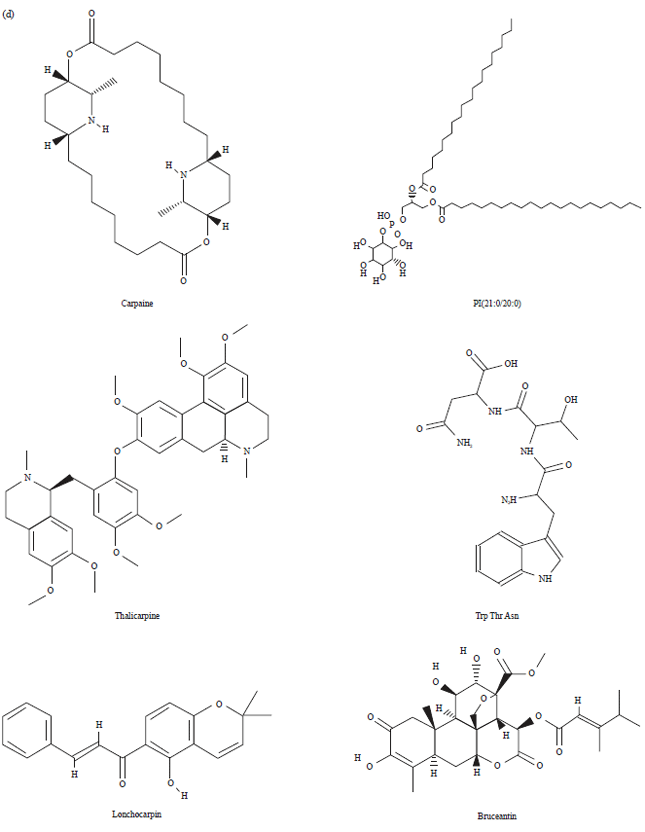
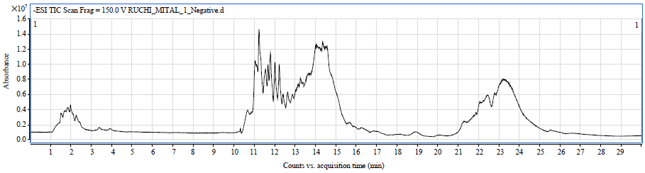
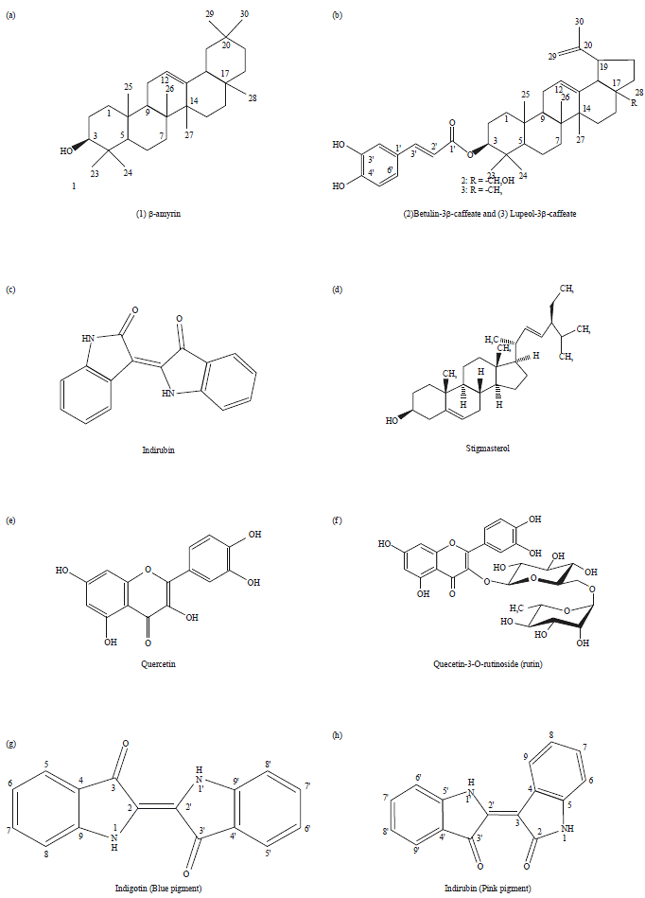
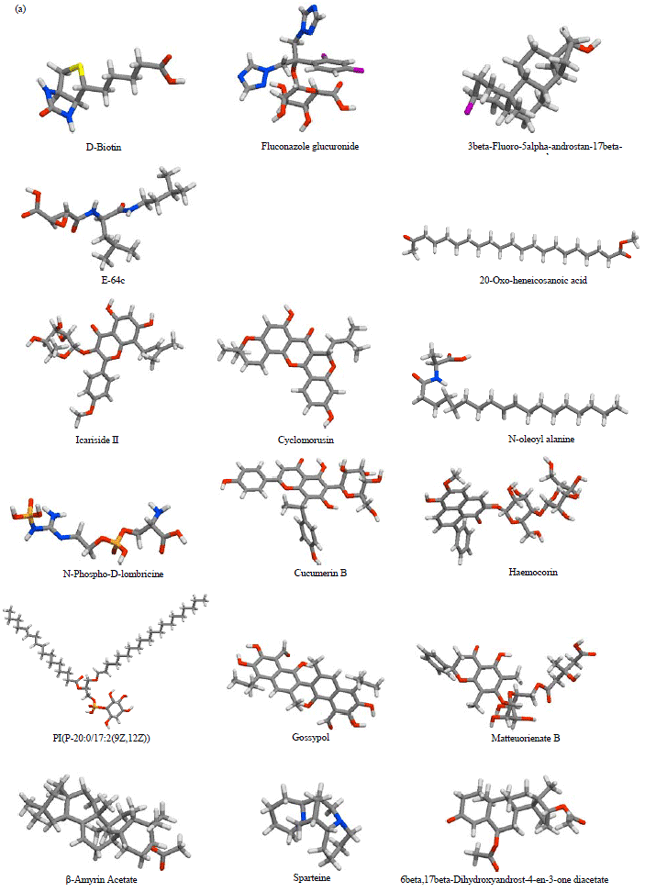
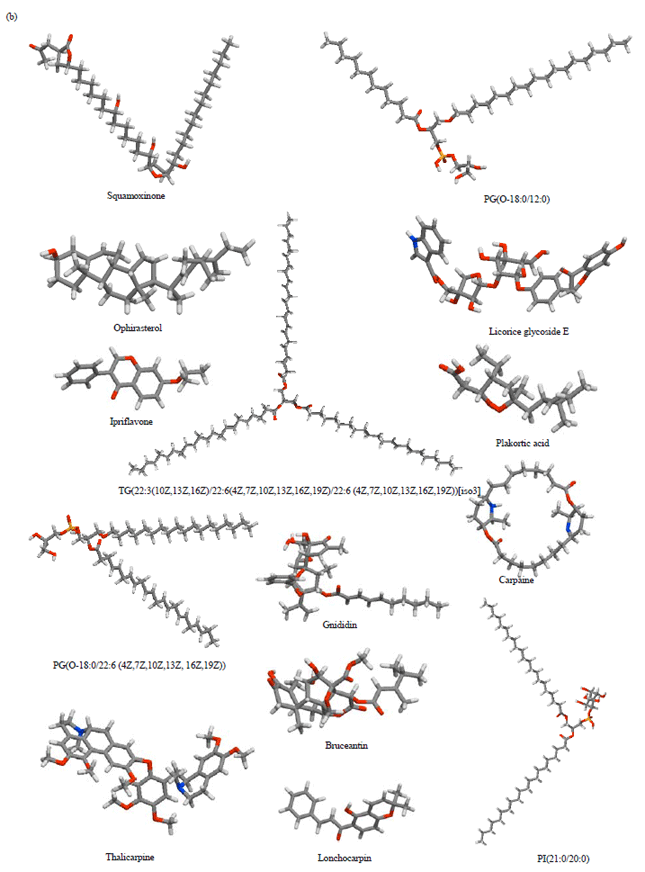
Research Article
Metabolite Profiling and Antioxidant Potency of Couroupita guianensis Aubl. Using LC-QTOF-MS Based Metabolomics
Department of Biosciences (UGC-CAS), Faculty of Science, Saurashtra University, 360005 Rajkot, Gujarat, India
LiveDNA: 91.8542
Kalpna Rakholiya
Main Dry Farming Research Station, Junagadh Agricultural University, Targhadia, 360003 Rajkot, Gujarat, India
LiveDNA: 91.7938
Ruchi Jakasania
Department of Biosciences (UGC-CAS), Faculty of Science, Saurashtra University, 360005 Rajkot, Gujarat, India
Rajesh Dave
Food Testing Laboratory, Department of Biotechnology, Junagadh Agricultural University, 362001 Junagadh, Gujarat, India
Sumitra Chanda
Department of Biosciences (UGC-CAS), Faculty of Science, Saurashtra University, 360005 Rajkot, Gujarat, India