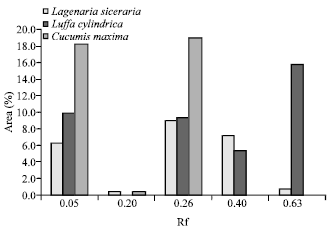
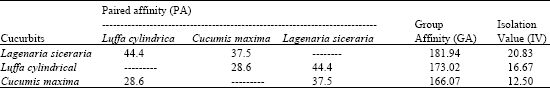
Research Article
Phytochemical Screening and High Performance TLC Analysis of Some Cucurbits
Department of Biosciences, Jamia Millia Islamia (Central University), New Delhi, 110025, India
Irfan Ahmad
Department of Biosciences, Jamia Millia Islamia (Central University), New Delhi, 110025, India
H.C. Goel
Department of Biosciences, Jamia Millia Islamia (Central University), New Delhi, 110025, India
M. Moshahid A. Rizvi
Department of Biosciences, Jamia Millia Islamia (Central University), New Delhi, 110025, India