Research Article
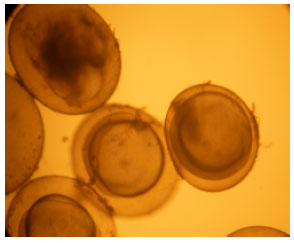
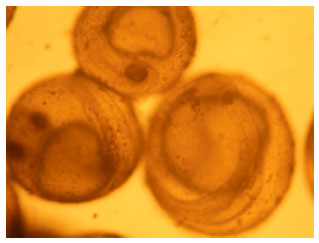
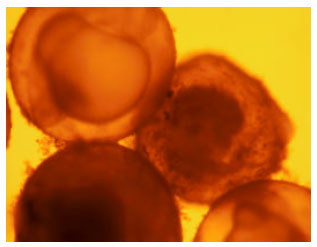
Impact of Paper Mill Effluent on the Survival and Hatchability of Eggs of Cyprinus carpio
Fish and Fisheries Laboratory, Department of Zoology, Kurukshetra University, Kurukshetra-136119, India
A. Fulia
Fish and Fisheries Laboratory, Department of Zoology, Kurukshetra University, Kurukshetra-136119, India
R.K. Sharma
Fish and Fisheries Laboratory, Department of Zoology, Kurukshetra University, Kurukshetra-136119, India