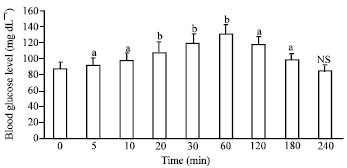
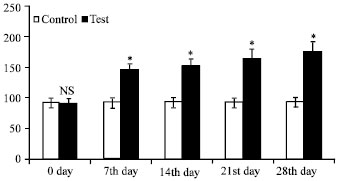
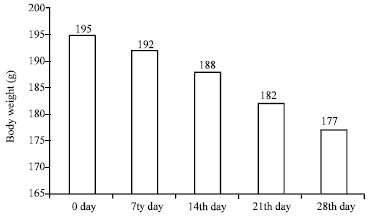
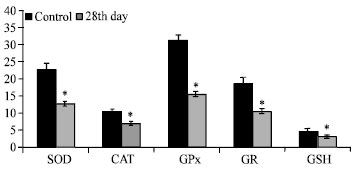
Research Article
Effects of Inhalational Exposure of Malathion on Blood Glucose and Antioxidants Level in Wistar Albino Rats
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
Pradeep G. Nayar
Department of Cardiology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
Venu Gopala Rao Konda
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
N. Madhusudhanan
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
E. Madhavi
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
M. Chokkalingam
Department of Cardiology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
Vinayak Meti
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India
S. Sundaravalli
Department of Pharmacology, Chettinad Hospitals and Research Institute, Kelambakkam, Chennai, India