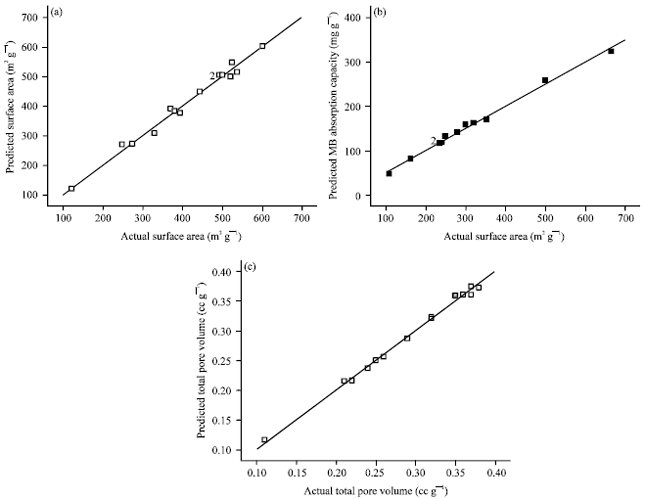
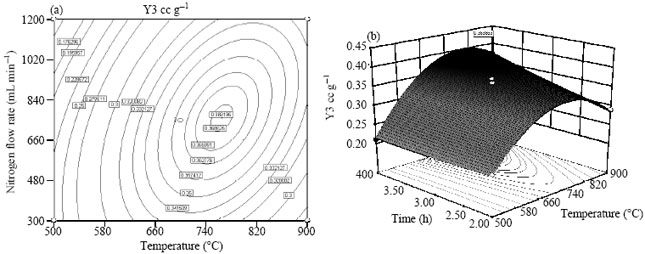
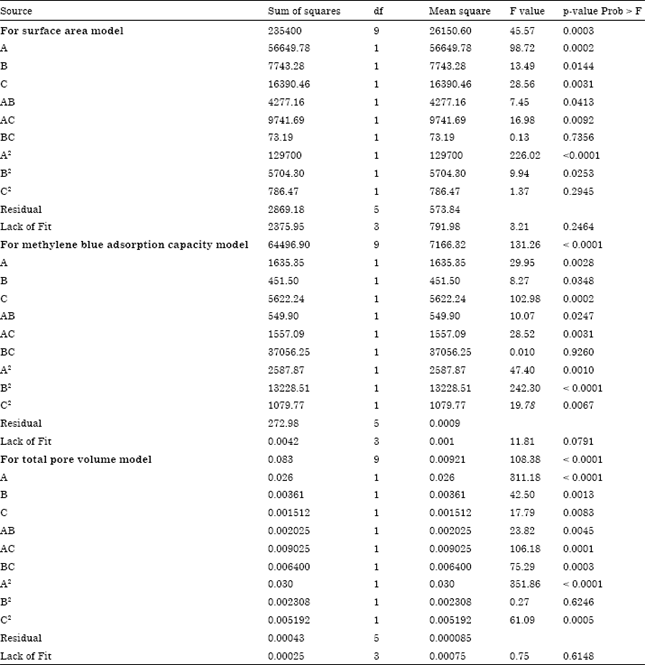
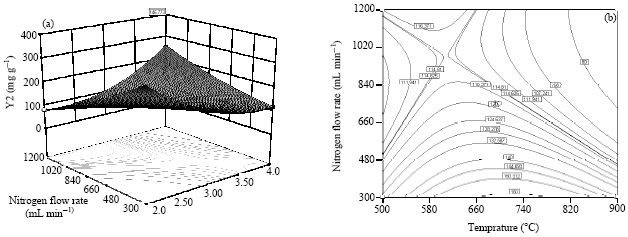
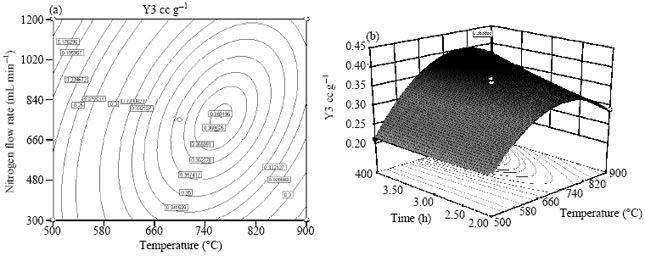
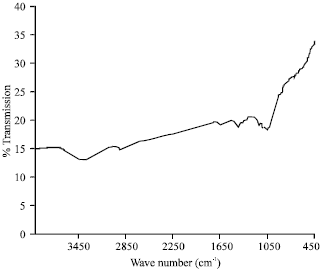
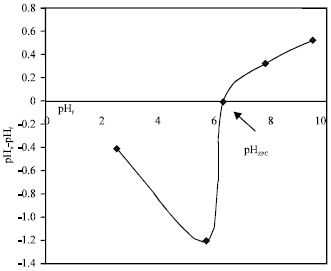
Research Article
Application of Response Surface Methodology for the Removal of Methylene Blue by Acacia Auriculiformis Scrap Wood Char
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, West Bengal-721302, India
Jayanta Kumar Basu
Department of Chemical Engineering, Indian Institute of Technology, Kharagpur, West Bengal-721302, India