Research Article
Cadmium-Induced Hepatotoxicity and Oxidative Stress in Rats: Protection by Selenium
Department of Zoology, Faculty of Science, King Saud University, P.O. Box 2455, Riyadh 11451, Saudi Arabia
Cadmium (Cd), a divalent metal toxicant, preferentially localized in hepatocytes and causes liver injury (Sumathi et al., 1996). Cd is well known for its various adverse effects, e.g., enhancement of lipid peroxidation (Manca et al., 1991), influence on mitochondrial function (Southard et al., 1974) and DNA chain break (Tsuzuki et al., 1994).
Selenium (Sel) is an important part of the antioxidant system of animals tissues (Burk and Hill, 1993). Selenium has previously been found to counteract the physiological effects of cadmium on male gonads (Alhazza, 2005). Moreover, Sel interacts with Cd in culture cells in relation to cellular uptake (Frisk et al., 2002). Sel reduced alterations in liver, heart and spleen peroxidation and essential metal levels (Yiin et al., 2000). It is suggested that preventive supplement with Sel can reduce injury to human hepatocyte caused by lipid peroxidation (Burk, 2002). This study was conduced to elucidate the response of liver physiology in rats to chronic treatment with Cd or Cd+Sel.
Animals
Ninety-six male Wister rats were used in the present investigation. The age of rates was six months and their weights ranged from 250 to 290 g. The animals were obtained from Animal House, Faculty of Pharmacy, King Saud University. The rats were maintained under standard laboratory condition (12 h light, temperature 23±1°C). They fed dry ration ad lib.
Chemicals
Cadmium as cadmium chloride (CdCl2) and Selenium as Sodium selenite (Na2SeO3) were purchased from Merck (Dormstadt, Germany). The chemicals used were of the highest purity.
Experimental Design
The rats were randomly divided into three groups of thirty-two animals each. The groups were: group 1 served as control and received the equivalent volume of tap water, group 2 injected (s.c.) with 2.5 mg kg-1 body weight of cadmium chloride (Coyle et al., 2000) in 0.1 mL saline and group 3 injected (s.c.) with Selenium at dose level 0.35 mg kg-1 body weight (Jamba et al., 1997) 1 h prior to Cd injection (2.5 mg kg-1). The animals were treated four times weekly for eight weeks. Eight blood samples from each group were collected after 2, 4, 6 and 8 weeks and plasma was separated. Eight animals from each group were sacrificed at the different time intervals and liver samples were kept frozen.
Biochemical Assays
Blood hydroperoxide levels were evaluated using free radical analytical system (IRAM, PARMA, Italy). The test is a colorimetric test that takes advantage of the ability of hydroperoxides to generate free radicals after reacting with some transitional metals. When buffered chromgenic substance is added, a coloured complex appears. The complex can be measured by a spectrophotometer.
Cd and zinc contents in liver cells were measured using atomic absorption spectrophotometry. Certified reference solutions were used to generate standard curves for each element.
ALT, AST, ALP and γ-GT activities were determined by kinetic methods using bio Merieux kits, France. Moreover, total bilirubin concentration in plasma was evaluated colorimetrically.
Statistical Analysis
The data were expressed as mean values±SE of 8 rats in each group and statistical difference between groups were assessed by student t-test.
Cd administered subcutaneously resulted in a significant increase (p<0.01) of hydroperoxide level in blood by 50, 70, 83 and 128% compared to control at 2, 4, 6 and 8 weeks post-treatment (Table 1). The hydroperoxide level in Sel+Cd group increased significantly (p<0.01) by 31, 39, 49 and 63%, respectively at the previous time intervals. It is evident that hydroperoxide level of rats treated with Sel and Cd was significantly (p<0.01) less than that of rats treated with Cd alone at the four time intervals. Hepatic Cd concentration (Table 1) showed a significant increase (p<0.01) in the treatment groups compared with control group at all time intervals. Cd concentration of Sel+Cd group was less than in Cd group.
Table 2 shows hepatic zinc concentration and AST activity in rats treated with Cd or Sel+Cd. Zinc concentration in liver of treatment groups increased significantly (p<0.01) compared with control group. AST activity increased significantly (p<0.01) in Cd treated group at all time intervals compared with control group, but increased significantly in Sel+Cd group at 8 weeks post treatment only.
ALT and ALP activities (Table 3) elevated significantly in the rats injected with Cd only compared with control group. ALT activity of Sel+Cd group increased significantly (p<0.01) after 8 weeks compared with control group, but ALP activity did not change significantly in the same group.
Table 4 showed that γ-GT activity and bilirubin concentration elevated with Cd only compared with control group, while they appear normal in rats given Sel and Cd in combination except γ-GT at 8 weeks post-treatment which increased significantly (p<0.01) compared to control.
| Table 1: | Effect of subcutaneous administration of Sel and Cd on blood hydroperoxide level and hepatic cadmium concentration in male rats |
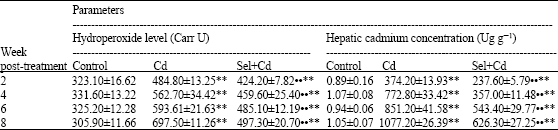 | |
| Values are means±SE (n = 8). ** Significantly different from control, p<0.01; •$ Significantly different from Cd group, p<0.01 | |
| Table 2: | Effect of subcutaneous administration of Sel and Cd on hepatic zinc concentration and blood AST activity in male rats |
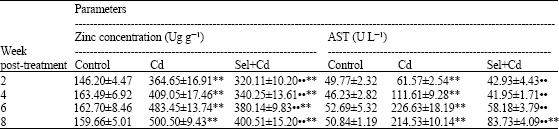 | |
| Values are means±SE (n = 8); ** Significantly different from control, p<0.01; •$ Significantly different from Cd group, p<0.01 | |
| Table 3: | Effect of subcutaneous administration of Sel and Cd on blood ALT and ALP activities in male rats |
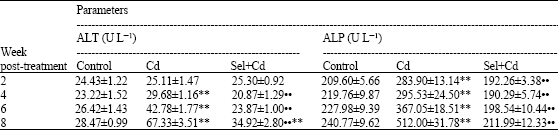 | |
| Values are means±SE (n = 8); ** Significantly different from control, p<0.01; •$ Significantly different from Cd group, p<0.01 | |
| Table 4: | Effect of subcutaneous administration of Sel and Cd on blood γ-GT and bilirubin in male rats |
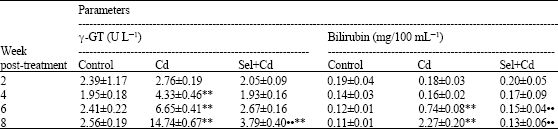 | |
| values are means±SE (n = 8). ** Significantly different from control, p<0.01. •$ Significantly different from Cd group, p<0.01 | |
The values of all previous parameters in Sel+Cd group are less than those of rats treated with Cd only.
Cadmium is perhaps one of the most toxic industrial and environmental metals and possess a continuing health hazard. Since it is rapidly distributed in the tissues (Waalkes et al., 1992).
The results of our experiments showed that Cd elevated hydroperoxide level. Similar results were recorded by Bashandy et al. (2006). The production of reactive oxygen species may associate with Cd toxicity. In vivo, Cd increases the formation of thiobarbituric acid reactive substances in lungs, liver and brain and urinary excretion of malondialdehyde, a product of lipid peroxidation (Farris, 1991; Manca et al., 1991; Muller, 1991). It has been proposed that the enhancement of lipid peroxidation by cadmium in rats is a consequence of a decrease of antioxidant enzymes (Jurczuk et al., 2004). Lipid peroxides that accumulate due to lipid peroxidation are known to be harmful to cells and tissue (Weiss and LoBuglio, 1982). The relation between the hepatic tissue damage and elevation of the liver enzymes is well documented (Sidhu et al., 2004). The observed increase in the activities of plasma AST, ALT, ALP and γ-GT is likely due to lipid peroxidation of biomembranes which causes leakage of cellular components (Matsu et al., 1989). It seems that, the increase of liver enzymes may be due to accumulation of Cd in hepatic tissue which enhances formation of lipid peroxidation. The present investigation showed that injection of Cd to rats increased hepatic zinc concentration. Cd induces changes in homeostasis of zinc resulting in an increased retention in zinc in the liver which is due to metallothionein induction in this organ (Brozoska et al., 2000).
Sel has been shown to protect against toxicity of various chemicals induced oxidative damage (Borges et al., 2006). The mechanism by which Sel acts has been primarily attributed to inhibition of oxidative stress induced by these chemicals or the formation of a complex between Sel and Cd (Santos et al., 2005).
Selenium reduced lipid peroxidation induced by Cd (Yiin et al., 2000). In the present study, selenium reduced the accumulation of Cd in the liver cells which may lead to a decrease in hydroperoxide level. The present experiments showed that Sel alleviated the deleterious effects of Cd on liver enzymes and bilirubin. The ameliorating effect of Sel on biochemical parameters (ALT, AST, GGT, ALP and bilirubin) might be due to an interaction of Sel with Cd, forming biologically inactive cadmium selenide complexes (Whanger et al., 1980) or due to decreased lipid peroxidation, antioxidant property and scavenging free radicals in liver (Miller et al., 2007) and increased hepatic glutathione content, a powerful antioxidant. Yuan and Tang (1999) reported that selenium is one of the necessary trace elements in the body which has the ability to counteract free radicals. Sel has antioxidant properties because of its presence in the active center of glutathione peroxidase (Rotruck et al., 1973). Sel might be acting through increasing the glutathione peroxidase activity and thus provides protection against oxidative stress (Imam et al., 1999). In conclusion, present results demonstrate that Cd induced changes in the physiology of liver and Sel reduced the deleterious effects of Cd.