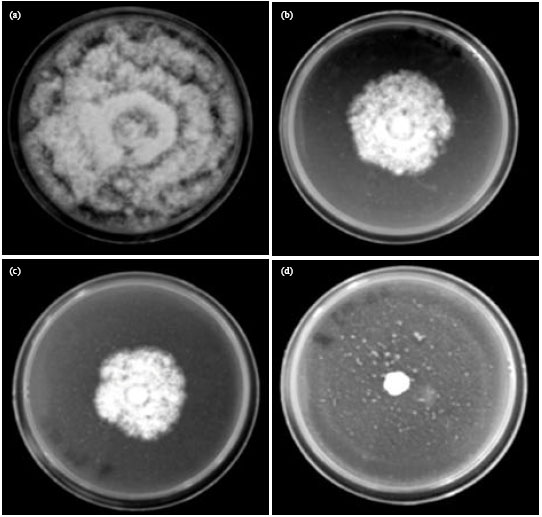
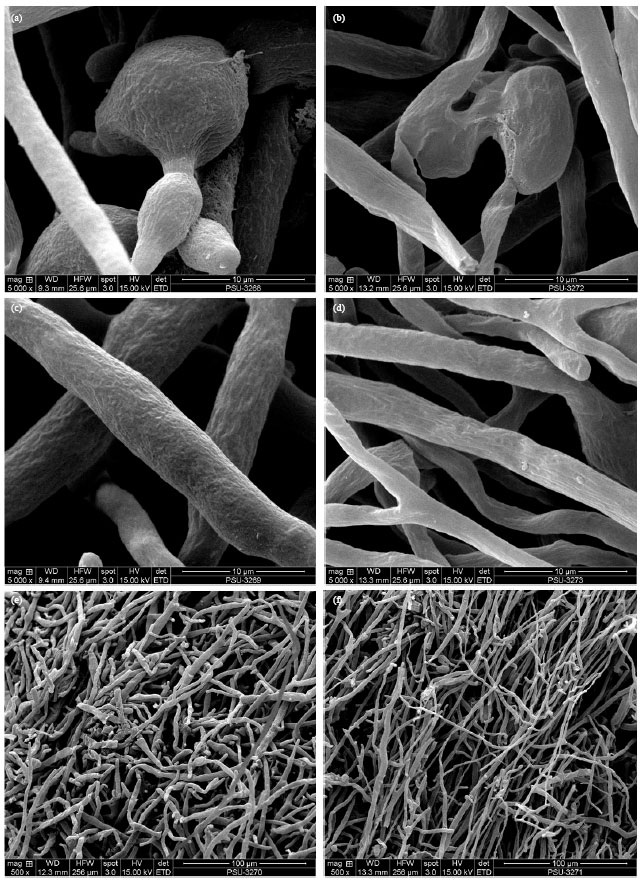
Research Article
Chitosan Inhibits the Growth of Phytophthora botryosa: The Causal Agent of Para Rubber Leaf Fall Disease
Department of Pest Management, Faculty of Natural Resources, Prince of Songkla University, Songkhla 90112, Thailand
Chaninun Pornsuriya
Department of Pest Management, Faculty of Natural Resources, Prince of Songkla University, Songkhla 90112, Thailand