Research Article
Biological Control of Damping Off and Root Rot of Wheat and Sugar Beet with Trichoderma harzianum
Department of Plant Pathology, Faculty of Agriculture, Assiut University, Assiut 71526, Egypt
INTRODUCTION
Wheat is considered an essential crop in human nutrition. Damping off and root rot diseases caused by Fusarium nivale, F. graminearum and F. tricinctum, these are an important diseases of wheat (Burmeister and Plattner, 1987; Bushnel et al., 2003; Bai and Shaner, 2004; Schaafsma and LincieI, 2005; Tunali et al., 2006a, b; Kohl et al., 2007; Nedelink et al., 2007; Chibundu et al., 2008; Gargouri-Kammoun et al., 2009). Sugar beet is considered as the second crop for sugar in Egypt. Damping off and root rot caused by F. lateritium, F. xylarioides and F. camptocearas is an important disease of sugar beet (El-Kazzaz et al., 2008). Herbicides and fungicides are environmental pollutants, therefore, using biological control such as T. harzianum which is one the efficient biocontrol agents that commercially produced to prevent development of several soil pathogenic fungi (Shalini et al., 2006; Ozbay and Newan, 2004; Osman et al., 2011; Yadav et al., 2011). Different mechanisms have been suggested as being responsible for their bio-control activity which includes mycoparasitism, antibiosis, competition for nutrients and space and secretion of chitinolytic enzymes (Harman, 2000). Chitinase 42 kDa produced by Trichoderma harzianum has been proven as a prime compound to be excreted into the hyphae of the pathogen causing localized cell wall lyses at the point of interaction (Muskhazli et al., 2008). The most effective biocontrol agent was Trichoderma harzianum. Hyphal interactions between T. harzianum and R. solani were observed by scanning electron microscopy. T. harzianum attached to the host by hyphal coils (Tarek and Moussa, 2002). Trichoderma harzianum not only prolonged the metabolic activity of the entrapped organism but also it promotes slow release of microbial spores into the medium for successful enzyme production (Attitalla and Salleh, 2010). The objective of the study was to show that disease reduced more in seed coating method than soil method.
Pathogens: F. nivale, F. graminearum and F. tricinctum were isolated from naturally infected wheat plants as well as F. Lateritium, F. xylarioides and F. camptocearas were isolated from naturally infected sugar beet plants and showing damping off and root rot symptoms on cultivated plants in Assiut Governorate, Egypt. Isolation technique was carried out according to Galal and Hefnawy (2002). Isolated fungi were purified using single spore and hyphal tip protein patterns technique and identified according to descriptions in the manual of the isolated fungi were identified on the basis of morphological and culture characteristic according to Nelson et al. (1983), Booth (1971), Moubasher (1993) and Leslie and Summerell (2006) and then confirmed by Assiut University Mycological Center (AUMC).
Trichoderma isolation: The microorganism was isolated from rhizosphere and bulk soil of wheat fields according to Elad and Chet (1983).
Inoculum preparation of antagonistic Trichoderma: To produce inoculum 80 g wheat seeds, 80 g sand and 80 mL distilled water for Trichoderma harzianum were separately added to 500 mL Erlenmeyer flask and sterilized twice at 121°C for 20 min, with a 3 day interval between autoclaving. Flasks containing autoclaved medium were inoculated with Trichoderma young colonies grown on 1/4-strength PDA, respectively and incubated in light at 21-25°C, until completely colonized 10 days for soil treatment application the prepared inoculum was aired, milled and incubated until application time in ice house (Duffy et al., 1997) to supply suspension of Trichoderma spore for seed coating method 20 mL distilled water was added to flasks containing Trichoderma inoculums (Not milled sporulated. Stock suspension was collected into 20 mL tubes and homogenized by vibrator. Quality of the inoculum the number of propagules per gram or milliliter inoculums was set 106 propagules per gram of inoculums for soil treatment and 107 conidia on seed surface for seed coating method using colony forming unit and haemocytometer laboratorial process, respectively.
Greenhouse tests: The effect of T. harzianum on the incidence of damping off of wheat and sugar beet disease was carried out under the greenhouse conditions at the Assiut University during growing seasons 2010 and 2011 Salty loam soil with 1% organic matters was used. Completely randomize of designs of seed coating and soil treatment methods were accomplished in greenhouse. Sterilized pots (25 cm in diameter) were filled with sterilized clay loam soil. The soil was artificially infested with tested fungi in individually by adding infested barley grains medium (Fahim et al., 1981) at the rate of 5% of soil weight and mixed well the pots were maintained under greenhouse. The surface sterilized Sads1 wheat seeds and Dempoloy sugar beet seeds were coated with (T. harzianum) at a rate of 107 conidia/averagely on seed surface for seed coating method.
In soil treatment method, contaminated soil was treated with inoculums of Trichoderma isolates averagely 106 propagules per gram at the rate of 10 g kg-1. Five wheat seeds or five sugar beet seeds were planted in each pot at 2 cm depth.
The percentage of pre and post emergence damping off, survival seedlings and disease index for seedlings were recorded after 21 and 45 days, respectively from planting. Disease index were recorded at the end of the experiment by using the scale of 0-4 used by (Achenbach and Jeunifer, 1996). At the end of the experiment the dry weight of shoot and root was.
Evaluation of antagonistic activity of Trichoderma species
In dual culture technique (in vitro): T. harzianum was carried out according to Coskuntuna and Ozer (2008) by using dual culture technique. F. nivale, F. graminearum, F. tricinctum, F. lateritium, F. xylarioides and F. camptocearas, separately, on PDA medium for 7 days at 25°C. Disc (5 mm-diameter) from each bio-control fungus was inoculated on surface of PDA medium in side of petri dish. A disc (5 mm-diameter) of F. nivale, F. graminearum, F. tricinctum, F. lateritium, F. xylarioides and F. camptocearas, separately was inoculated at equal distance of the opposite side of petri dish. Petri dishes were inoculated with each pathogenic fungus only as control. Three Petri dishes for each bio-control-pathogenic fungus treatment as well as the control were used as replicates. The inoculated Petri dishes were incubated at 25°C at 7 days when the pathogen fungi covered the plate surface of the control treatment, then T. harzianum and pathogens were evaluated based on radial growth of colony of pathogen, over growth of Trichoderma.
Antagonistic effect of T. harzianum as decrease of the mycelia growth of pathogenic fungi was determined using the following formula:
Where:
| A | = | The diameter of mycelia growth of pathogenic fungus in control |
| B | = | The diameter of mycelia growth of pathogenic fungus with Trichoderma fungus |
Culture filtrate (nonvolatile metabolites) and early volatile metabolites tests: Mycelia disks of each Trichoderma isolate grew on 1/4-strength PDA was separately inoculated into 100 mL flasks containing potato dextrose liquid and incubated at 20-29°C and 120 RPM in rotary shaker incubator for 10 days. The cultures were then filtered through 0.22 mm Millipore filters and 15 mL of these filtrates were added into sterile Erlenmeyer flasks containing 50 mL 1/4-strength PDA with 25% further agar at 45°C. After medium solidifying, mycelia disks of F. nivale, F. graminearum, F. tricinctum, F. lateritium, F. xylarioides and F. camptocearas individually agent derived from actively growing colonies were placed on one edge of medium plates and were incubated at 25±3°C (Dennis, 1971; Kucuk and Kivanc, 2003, 2004). For early volatile metabolites test, pathogen and Trichoderma actively growing colonies were subculture on PDA and incubated in dark condition at 25°C. Then, opened Petri dishes containing 48 h old colony agent F. nivale, F. graminearum, F. tricinctum, F. lateritium, F. xylarioides and F. camptocearas placed on 24 h old colony of Trichoderma and were airtight using parafilm. Control was Petri dishes containing PDA medium. The Petri dishes were incubated in the same temperature and dark conditions (Dennis, 1971; Fiddaman and Rossall, 1993). Radial growth on pathogen was measured daily in both tests. Inhibitory percentages were calculated by above formula.
Statistical analysis: Data were subjected to statistical analysis and means were compared using LSD at test (Gomez and Gomez, 1984).
In vitro antagonistic effect of T. harzianum against the cause of wheat and sugar beet damping off disease: Dual culture assays provided evidence that T. harzianum substantially reduced the growth of F. nivale, F. graminearum and F. tricinctum the causal pathogens of wheat as well as F. lateritium, F. xylarioides and F. camptoceras the causal pathogen of sugar beet compared with the control. The Trichoderma harzianum grew over and sporulated of the different Fusarium spp. resulting of complete degradation (Table 1-2).
Data also indicate that the T. harzianum and its filtrate inhibited the growth of the pathogens (F. nivale, F. graminearum, F. tricinctum, F. lateritium, F. xylarioides and F. camptoceras) (Table 3-4).
F. xylarioides and F. graminearum showed the highest percentage of growth reduction while the F. lateritium and F. camptoceras showed the lowest percentage of growth reduction.
Data also indicate that filtrate of T. harzianum significant different inhibited between (F. nivale, F. graminearum, F. tricinctum) the causal pathogen of damping off wheat. Data also indicate that filtrate of T. harzianum significant different inhibited between F. lateritium, F. xylarioides and F. camptoceras) the causal pathogen of damping off sugar beet.
Antagonistic effect of T. harzianum against the causative pathogen of wheat and sugar-beet damping off and root rot disease under greenhouse conditions: Coating wheat seeds with fungal bioagent or soil treatment with fungal bioagent resulted in different degree of protection against the causal pathogen at the seedling stage.
| Table 1: | Reaction and antifungal of Trichoderma harzianum on the causative agent of wheat damping off and root rot in vitro |
 | |
| Table 2: | Reaction and antifungal of Trichoderma harzianum on the causative agent of sugar beet damping and root rot in vitro |
 | |
| Table 3: | Effect of culture filtrates of T. harzianum on growth on the causative agent of wheat damping-off and root rot |
 | |
| Table 4: | Effect of culture filtrates of T. harzianum on growth on the causative agent of sugar beet damping-off and root rot |
 | |
Minimal amount of disease were observed on plants inoculated with pathogen and bioagent compared with untreated control. Science T. harzianum reduced the percentages of disease incidence and disease severity of Fusarium spp. the causal pathogen of wheat and sugar beet compared with control.
The highest percentage of seedling survival, shoot dry weight, root rot weight and the least disease severity were associated with F. graminearum and F. lateritium in two growing seasons but the lowest percentage of seedling survival, shoot dry weight, root rot weight and highest disease severity were associated with F. tricinctum and F. camptoceras in tested seeds treatment. Seed coating method was more effective than soil treatment method. Seed and soil treatment decreased seedling damping off and increased the average of shoot dry weight and root dry weight of wheat and sugar beet compared with the untreated and control). Data also indicate that T. harzianum reduce the percentages of pre not significantly caused by Fusarium nivale F. graminearum and F. tricinctum during growing two seasons. Data also indicate that T. harzianum reduce the percentages of disease severity significantly caused by Fusarium nivale, F. graminearum and F. tricinctum.
| Table 5: | Effect of T. harzianum on disease incidence and disease severity of damping off and root rot of wheat and dry weight of shoot and root under green house condition during growing season 2010 |
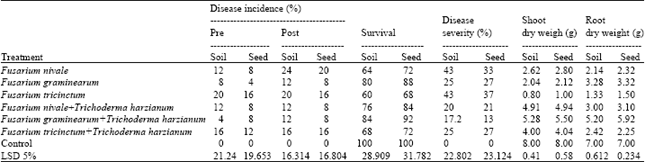 | |
| Table 6: | Effect of T. harzianum on disease incidence and disease severity of damping off and root rot of wheat and dry weight of shoot and root under green house condition during growing season 2011 |
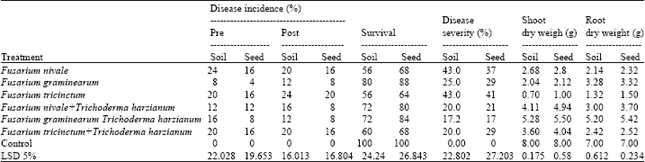 | |
| Table 7: | Effect of T. harzianum on disease incidence and disease severity of damping off and root rot of sugar beet and dry weight of shoot and root under green house condition during growing season 2010 |
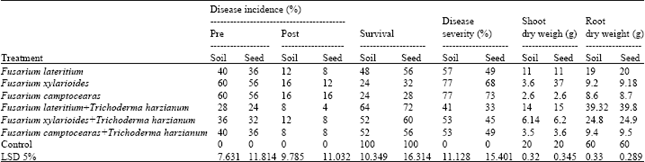 | |
| Table 8: | Effect of T. harzianum on disease incidence and disease severity of damping off and root rot of sugar beet and dry weight of shoot and root under green house condition during growing season 2011 |
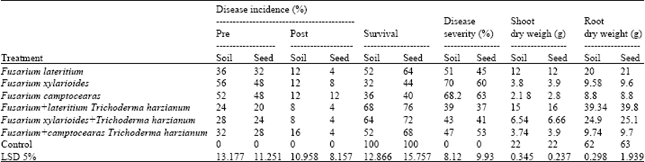 | |
Control untreated during growing two seasons Data also indicate that T. harzianum reduce the percentages of pre and the percentages of disease severity significantly caused by F. lateritium, F. xylarioides and F. camptoceras. Data also indicate that T. harzianum increase shoot dry weight, root rot weight significantly of wheat and sugar beet compared with the untreated and control during growing two seasons (Table 5-8).
T. harzianum has been reported as the best antagonists for damping off disease caused by Fusarium spp. under laboratory conditions. T. harzianum completely overgrew on the colony of the pathogens fungi. Trichoderma sp. have been previously demonstrated (Howell, 2003; Harman et al., 2004; Harman, 2006). T. harzianum treatment reduced the mycelia growth of the pathogenic fungi due to the rapid growth of T. harzianum which colonized medium surface and substrate. These observations are similar to Kucuk and Kivan (2004) and Mir et al. (2011).
Results also indicated that Trichoderma sp. reduced the disease incidence at pre and post emergence stage in pots. These results agree with those recorded by Kohl et al. (2007), El-Meleigi et al. (2007), Hajieghrari et al. (2008) and Siameto et al. (2010).
Results based on soil treatment with the T. harzianum demonstrated reduction to incidence of damping off and root rot disease in wheat and sugar beet under pot conditions (Riunge et al., 2007; Kohl et al., 2007; El-Meleigi et al., 2007; Hajieghrari et al., 2008; Siameto et al., 2010).
Treatment with Trichoderma sp. gave the highly protection to wheat and sugar beet seedlings against damping of disease at post emergence stage compared with pre emergence. It is may be related to the ability of Trichoderma strains is mycoparasitism mediated by the production of chitinases and other cell wall-degrading enzymes (Chet, 1987; Lorito et al., 1996).
Treating seeds and soil with T. harzianum greatly increased the average of shoot dry weight and root dry weight of wheat and sugar beet as compared with the control (Zafari et al., 2008).
Trichoderma harzianum may be very useful fungi in biological control against cereal aggressive and toxigenic F. species preventing Fusarium mycotoxin accumulation in plant tissues my results indicated that the potential of using of Trichoderma competitors for the control of mycotoxin production in grain as well as to reduce the toxigenic Fusarium inoculums levels in cereal debris it is considered a biocontrol strategy to reduce DON levels in crops residues as previously recorded by Busko et al. (2007).
It is concluded that Trichoderma species may be very useful fungi in biological control against wheat and toxigenic Fusarium species to reduce their inoculums and to prevent Fusarium mycotoxin accumulation in plant tissues.