Research Article
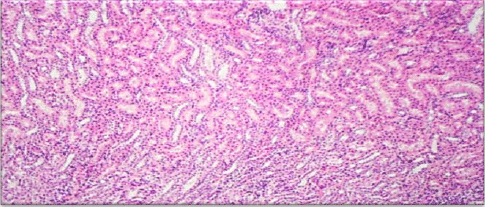
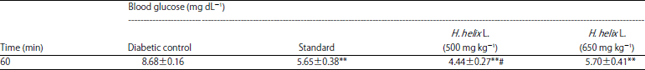
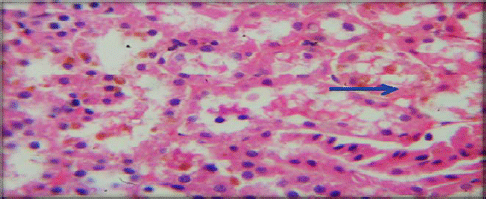
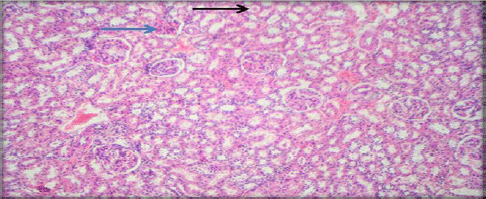
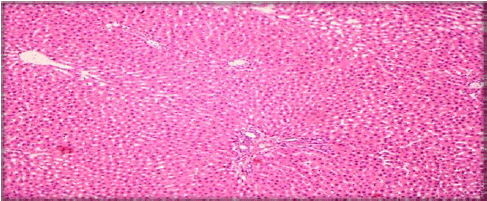
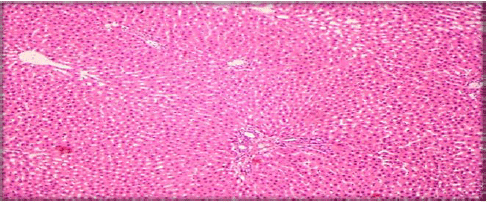
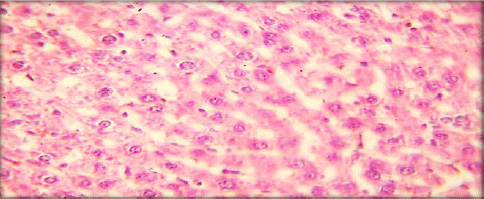
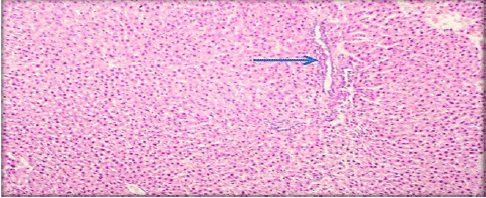
Evaluation of Acute and Chronic Antidiabetic Activity of Ivy (Hedera helix L.) Aqueous Leaf Extract in Rat Model
Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, University of Karachi, Karachi, Pakistan
Anum Adil
Department of Pharmacology, Faculty of Pharmacy and Pharmaceutical Sciences, University of Karachi, Karachi, Pakistan
Sadaf Naeem
Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University, Karachi, Pakistan
LiveDNA: 92.30093
Nazish Jaffar
Sindh Medical College, Jinnah Sindh Medical University, Karachi, Pakistan
Humera Khatoon
Jinnah University for Women, Karachi, Pakistan
Humera Ansar
Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University, Karachi, Pakistan
Yousra Shafiq
Institute of Pharmaceutical Sciences, Jinnah Sindh Medical University, Karachi, Pakistan