Research Article
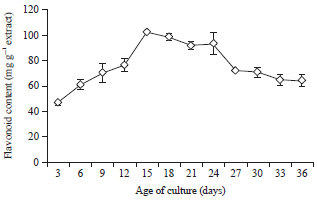
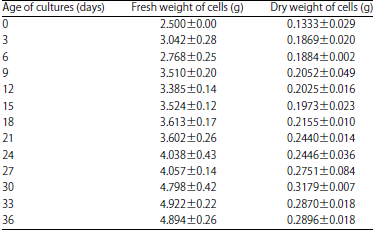
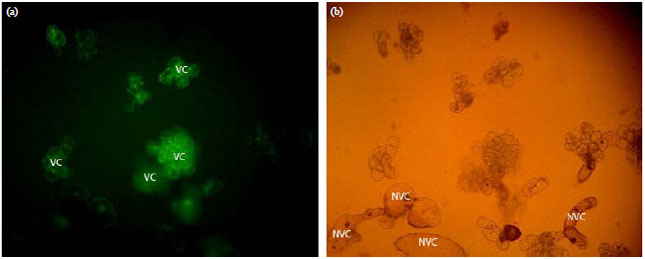
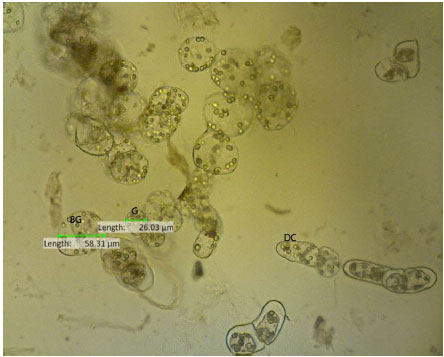
Flavonoid Production, Growth and Differentiation of Stelechocarpus burahol (Bl.) Hook. F. and Th. Cell Suspension Culture
Faculty of Biology, Universitas Gadjah Mada, 55281 Yogyakarta, Indonesia
LiveDNA: 62.14305
Sukarti Moeljopawiro
Laboratory of Biochemistry, Faculty of Biology, Universitas Gadjah Mada, 55281 Yogyakarta, Indonesia
LiveDNA: 62.14307
Kumala Dewi
Laboratory of Plant Physiology, Faculty of Biology, Universitas Gadjah Mada, 55281 Yogyakarta, Indonesia
LiveDNA: 62.14003
Ari Indrianto
Laboratory of Biotechnology, Faculty of Biology, Universitas Gadjah Mada, 55281 Yogyakarta, Indonesia
LiveDNA: 62.14320