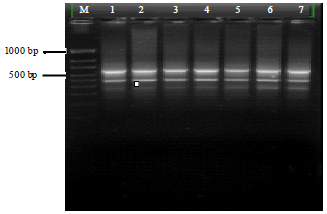
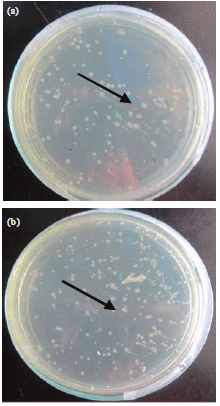
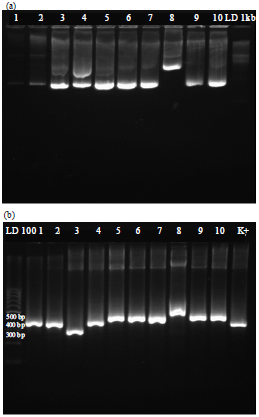
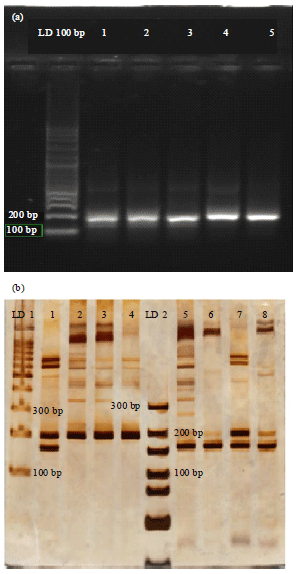
Research Article
Isolation and Characterization of Aedes aegypti Microsatellite Markers Dengue Hemorrhagic Fever Vector in West Sumatra, Indonesia
Subdivision of Parasitology, Faculty of Medicine, Alas University, Padang, Indonesia
Jamsari
Department of Agroecotechnology, Faculty of Agriculture, Alas University, Padang, Indonesia
Yanwirasti
Anatomy Subdivision, Faculty of Medicine, Alas University, Padang, Indonesia
Nuzulia Irawati
Subdivision of Parasitology, Faculty of Medicine, Alas University, Padang, Indonesia
Dahelmi
Department of Biology, Faculty of Mathematics and Natural Sciences, Alas University, Padang, Indonesia