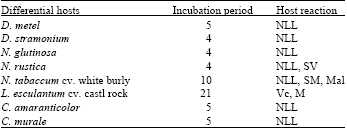
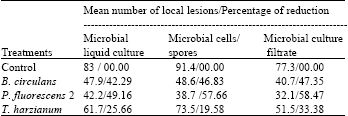
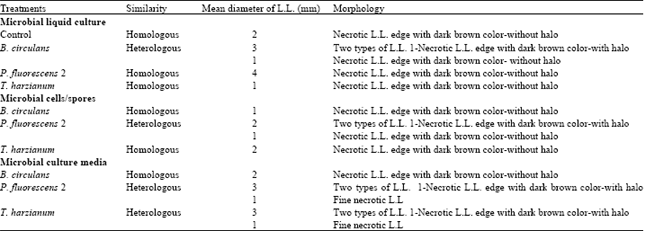
Research Article
Induction of Resistance in Tomato Plants Against Tomato mosaic tobamovirus Using Beneficial Microbial Isolates
Department of Plant pathology, National Research Centre, Dokki, Cairo, Egypt
Kh. A. El-Dougdoug
Department of Agriculture Microbiology, Faculty of Agriculture, Ain Shams University, Shoubra EL-Kheima, Cairo, Egypt
B.A. Othman
Department of Agriculture Microbiology, Faculty of Agriculture, Ain Shams University, Shoubra EL-Kheima, Cairo, Egypt
S.M. Lashin
Department of Plant pathology, National Research Centre, Dokki, Cairo, Egypt
M.A. Ibrahim
Department of Molecular Biology, Genetic Engineering and Biotechnology Division, National Research Centre, Dokki, Cairo, Egypt
A.R. Sofy
Department of Botany and Microbiology, Faculty of Science, Al-Azhar University, 11884 Nasr City, Cairo, Egypt