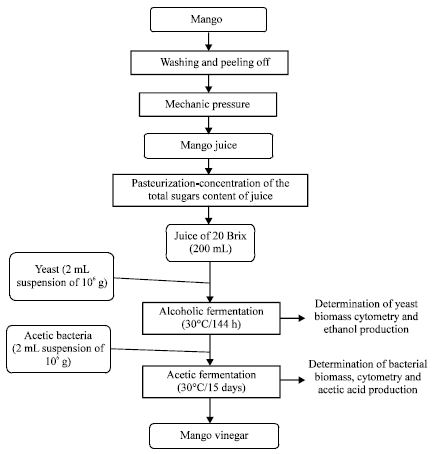
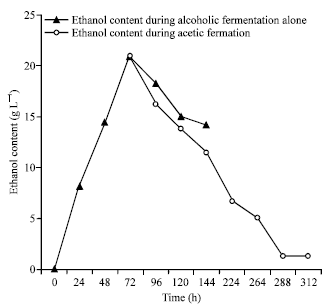
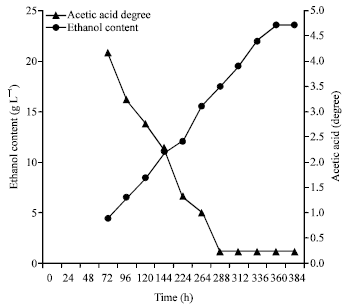
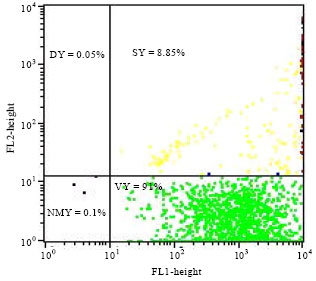
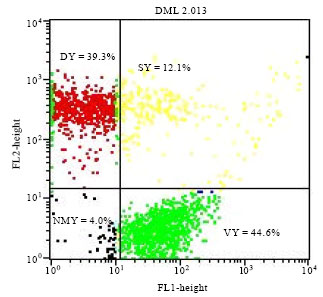
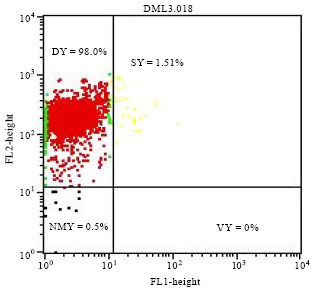
Research Article
Vinegar Production from Togolese Local Variety Mangovi of Mango Mangifera indica Linn. (Anacardiaceae)
Laboratoire de Microbiologie et de Contr�le de Qualite des Denrees Alimentaires, ESTBA-UL, Universite de Lome, Togo
Jean-Yves Leveau
Laboratoire de Microbiologie Industrielle, ENSIA, France
Simplice D. Karou
Laboratoire de Microbiologie et de Contr�le de Qualite des Denrees Alimentaires, ESTBA-UL, Universite de Lome, Togo
M. Bouix
Laboratoire de Microbiologie Industrielle, ENSIA, France
Seyram K. Sossou
Laboratoire de Microbiologie et de Contr�le de Qualite des Denrees Alimentaires, ESTBA-UL, Universite de Lome, Togo
C. De Souza
Laboratoire de Microbiologie et de Contr�le de Qualite des Denrees Alimentaires, ESTBA-UL, Universite de Lome, Togo