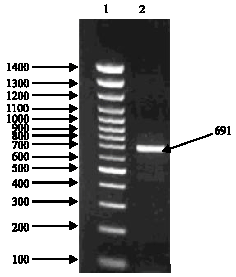
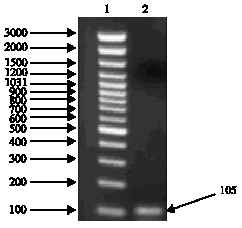
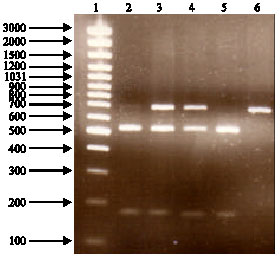
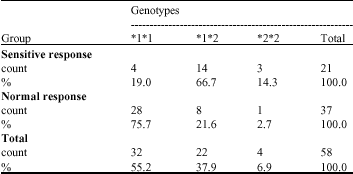
Research Article
CYP2C9 Gene Analysis of Some Iranian Hypersensitive Patients to Warfarin
Department of Biology, Faculty of Basic Sciences, Alzahra University, Tehran, Iran
R. Sharifian
Department of Hematology and Oncology, Tehran University, Tehran, Iran
Z. Jomeh Farsangi
Department of Biology, Faculty of Basic Sciences, Alzahra University, Tehran, Iran
Z. Kianmehr
Department of Biology, Faculty of Basic Sciences, Alzahra University, Tehran, Iran
M. Lak
Department of Hematology and Oncology, Tehran University, Tehran, Iran