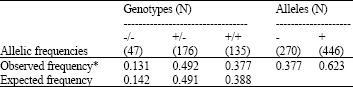
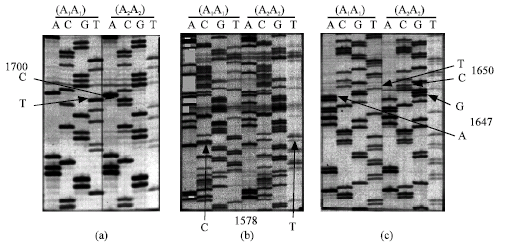
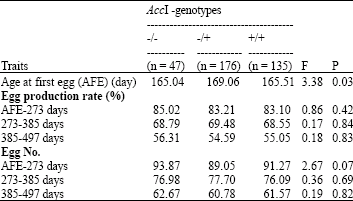
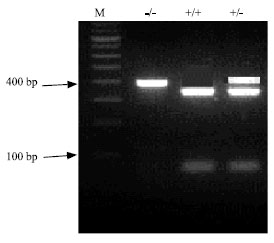
Research Article
Restriction Fragment Length and Single Strand Conformational Polymorphisms in Chicken Mitochondrial Phosphoenol-Pyruate Carboxykinase Gene and its Association with Egg Production
University of Sistan and Baluchistan, Zahedan, Iran
Urs Kuhnlein
Department of Animal Science, McGill University, Quebec, Canada