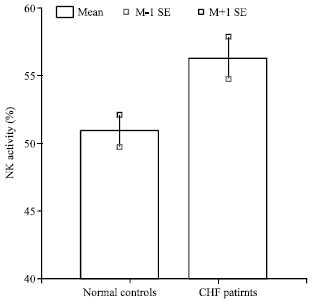
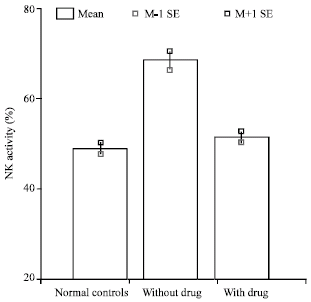
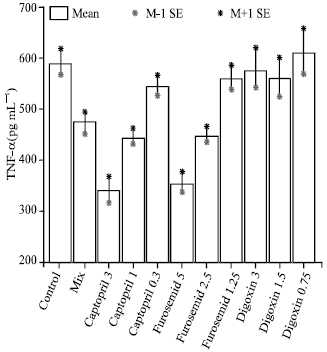
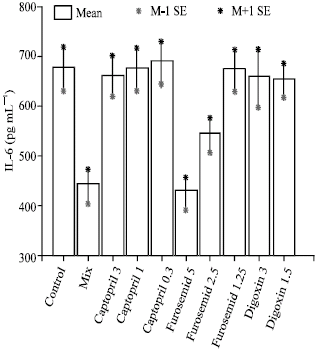
Research Article
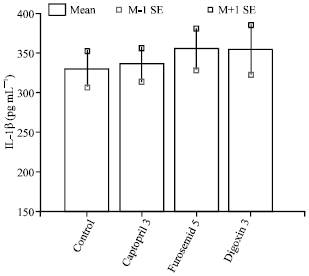
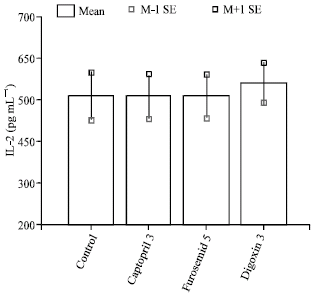
The Effect of Cardiovascular Drugs on Pro-Inflammatory Cytokine Secretion and Natural Killer Activity of Peripheral Blood Mononuclear Cells of Patients with Chronic Heart Failure in vitro
Department of Immunology,
Yahya Jaberi
Department of Cardiology, School of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran
Abdolreza Esmaeilzadeh
Department of Immunology, School of Medicine, Tarbiat Modarres University, Tehran, Iran
Mohammad Khani
Department of Cardiology, School of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran
Mehdi Moosaeefard
School of Nursing, Zanjan University of Medical Sciences, Zanjan, Iran
Mostafa Shafaqatian
Department of Immunology,