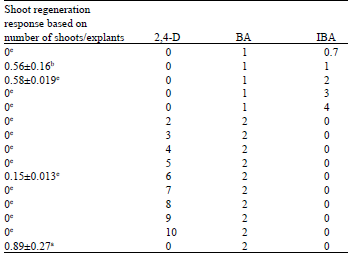
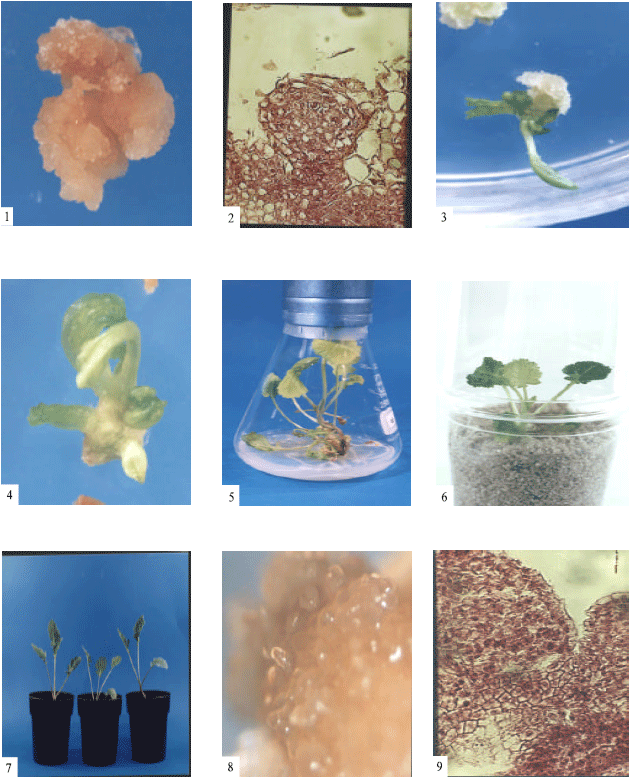
Research Article
In vitro Plant Regeneration from Callus of Cotyledons in Canola (Brassica napus L.)
Department of Biology, Faculty of Science, Teacher Training University of Tehran. No. 49, Mofatteh Ave, Tehran, Iran
A. Majd
Department of Biology, Faculty of Science, Teacher Training University of Tehran. No. 49, Mofatteh Ave, Tehran, Iran
S. Mehrabian
Department of Biology, Faculty of Science, Teacher Training University of Tehran. No. 49, Mofatteh Ave, Tehran, Iran