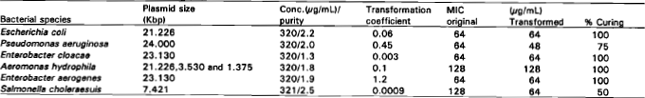
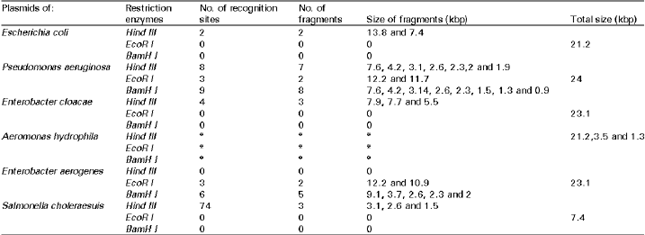
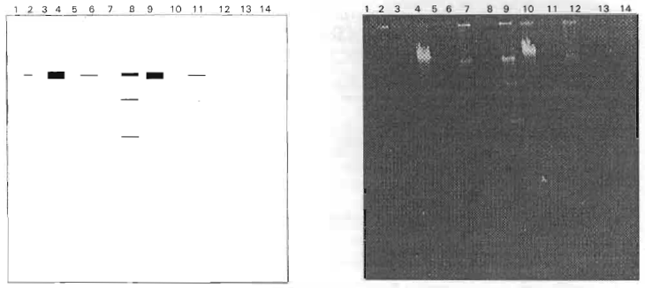
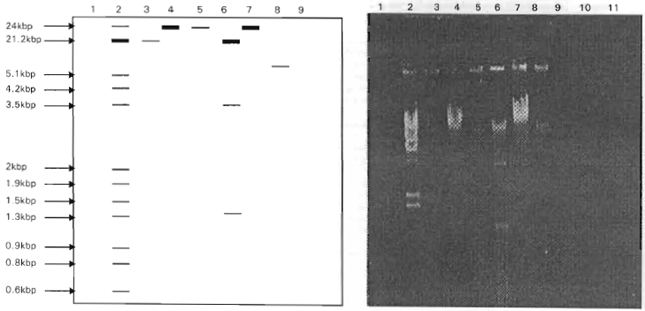
Research Article
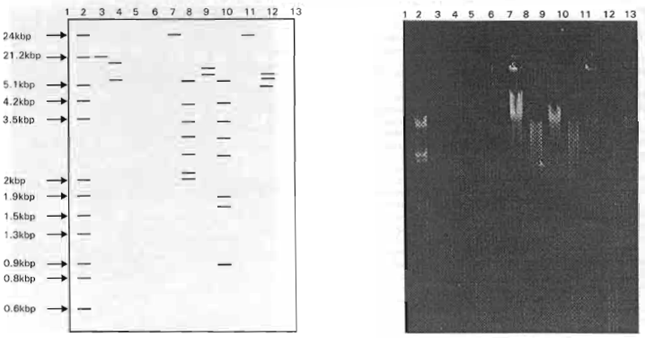
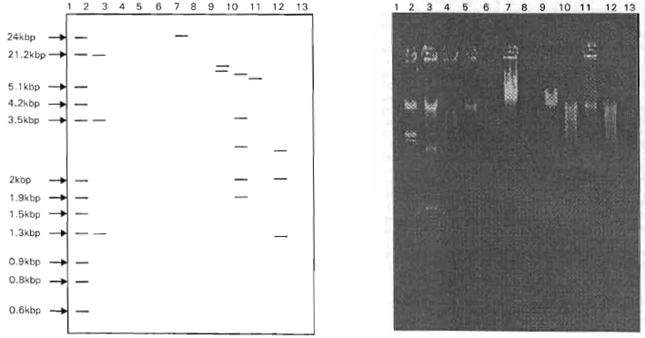
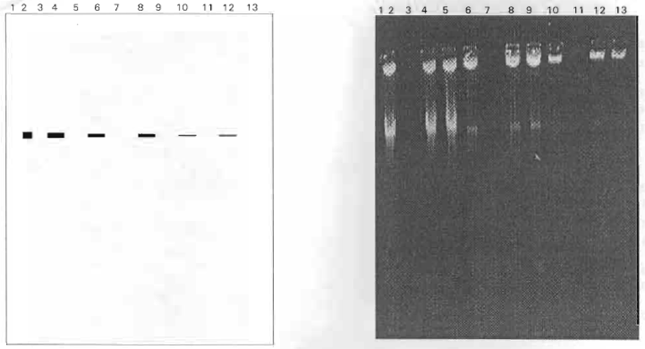
Plasmid-Encoded Transferable Antibiotic Resistance in Gram-negative Bacteria, Isolated from Drinking Water in Ismailia City
Department of Botany, Microbiology Section, Faculty of Science, Suez Canal University, Ismailia, Egypt
M.H. Abdel Aziz
Department of Botany, Microbiology Section, Faculty of Science, Suez Canal University, Ismailia, Egypt
S. A. Selim
Department of Botany, Microbiology Section, Faculty of Science, Suez Canal University, Ismailia, Egypt